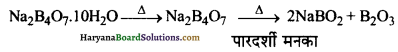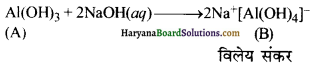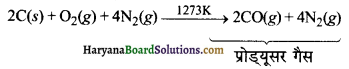Haryana State Board HBSE 11th Class Chemistry Solutions Chapter 11 p-ब्लॉक तत्त्व Textbook Exercise Questions and Answers.
Haryana Board 11th Class Chemistry Solutions Chapter 11 p-ब्लॉक तत्त्व
प्रश्न 1.
(क) B से Tl तक तथा (ख) C से Pb तक की ऑक्सीकरण अवस्थाओं की भिन्नता के क्रम की व्याख्या कीजिए।
उत्तर:
(क) B से Tl तक (बोरॉन परिवार) ऑक्सीकरण अवस्था [Oxidation state from B to Tl (Boron family)]—बोरॉन परिवार (वर्ग 13) के तत्वों का इलेक्ट्रॉनिक विन्यास ns2p1 होता है। इसका तात्पर्य यह है कि बंध निर्माण के लिए तीन संयोजी इलेक्ट्रॉन उपलब्ध हैं। इन इलेक्ट्रॉनों का त्याग करके ये परमाणु अपने यौगिकों में +3 ऑक्सीकरण अवस्था प्रदर्शित करते हैं। यद्यपि इन तत्वों की ऑक्सीकरण-अवस्था में निम्नलिखित प्रवृत्ति प्रेक्षित होती है-
(i) प्रथम दो तत्व बोरॉन तथा ऐलुमिनियम यौगिकों में केवल +3 ऑक्सीकरण अवस्था प्रदर्शित करते हैं, परन्तु शेष तत्व-गैलियम, इण्डियम तथा थैलियम +3 ऑक्सीकरण अवस्था के साथ-साथ +1 ऑक्सीकरण अवस्था भी प्रदर्शित करते हैं अर्थात् ये परिवर्ती ऑक्सीकरण अवस्थाएँ प्रदर्शित करते हैं।
(ii) +3 ऑक्सीकरण अवस्था का स्थायित्व ऐलुमिनियम से आगे जाने पर घटता है तथा अन्तिम तत्व थैलियम की स्थिति में, +1 ऑक्सीकरण अवस्था, +3 ऑक्सीकरण अवस्था से अधिक स्थायी होती है। इसका अर्थ यह है कि TlCI, TlCl3 से अधिक स्थायी होता है।
(ख) C से Pb तक (कार्बन परिवार) ऑक्सीकरण अवस्था [Oxidation state from C to Pb (Carbon family)]—कार्बन परिवार (समूह-14)के तत्वों का इलेक्ट्रॉनिक विन्यास ns2 np2 होता है। स्पष्ट है कि इन तत्वों के परमाणुओं के बाह्यतम कोश में चार इलेक्ट्रॉन होते हैं। इन तत्वों द्वारा सामान्यत: +4 तथा +2 ऑक्सीकरण अवस्था दर्शाई जाती है। कार्बन ऋणात्मक ऑक्सीकरण अवस्था भी प्रदर्शित करता है। चूँकि प्रथम चार आयनन एन्थैल्पी का योग अति उच्च होता है; अत: + 4 ऑक्सीकरण अवस्था में अधिकतर यौगिक सहसंयोजक प्रकृति के होते हैं।
इस समूह के गुरुतर तत्वों में Ge < Sn < Pb क्रम में +2 ऑक्सीकरण अवस्था प्रदर्शित करने की प्रवृत्ति बढ़ती जाती है। ऐसा सहसंयोजक कोश में ns2 इलेक्ट्रॉन के बन्धन में भाग नहीं लेने के कारण यह होता है।
इन दो ऑक्सीकरण अवस्थाओं का सापेक्षिक स्थायित्व वर्ग में परिवर्तन होता है। कार्बन तथा सिलिकन मुख्यतः +4 ऑक्सीकरण अवस्था प्रदर्शित करते हैं। जर्मेनियम की +4 ऑक्सीकरण अवस्था स्थायी होती है, जबकि कुछ यौगिकों में +2 ऑक्सीकरण अवस्था भी मिलती है। टिन ऐसी दोनों अवस्थाओं में यौगिक बनाता है (+2 ऑक्सीकरण अवस्था में टिन अपचायक के रूप में कार्य करता है)। +2 ऑक्सीकरण अवस्था में लेड के यौगिक स्थायी होते हैं, जबकि इसकी +4 अवस्था प्रबल ऑक्सीकारक है। इस आधार पर स्पष्ट है कि-
(i) SnCl4 तथा PbCl4 की तुलना में SnCl2 तथा PbCl2 अधिक सरलता से बनते हैं।
(ii) PbCl2, SnCl2 से अधिक स्थायी होता है चूँकि इसमें अक्रिय युग्म प्रभाव का परिमाण अधिक होता है।
चतुर्संयोजी अवस्था में अणु के केन्द्रीय परमाणु पर आठ इलेक्ट्रॉन होते हैं। इलेक्ट्रॉन परिपूर्ण अणु होने के कारण सामान्यतया इलेक्ट्रॉन ग्राही या इलेक्ट्रॉनदाता स्पीशीज की अपेक्षा इनसे नहीं की जाती है। यद्यपि कार्बन अपनी सहसंयोजकता +4 का अतिक्रमण नहीं कर सकता है, परन्तु समूह के अन्य तत्व ऐसा करते हैं। यह उन तत्वों में d-कक्षकों की उपस्थित के कारण होता है। यही कारण है कि ऐसे तत्वों के हैलाइड जल-अपघटन के उपरान्त दाता स्पीशीज (donor species) से इलेक्ट्रॉन ग्रहण करके संकुल बनाते हैं। उदाहरणार्थ-कुछ स्पीशीज; जैसे-
\(\left(\mathrm{SiF}_6\right)^{2-}\), \(\left(\mathrm{GeCl}_6\right)^{2-}\) तथा \(\mathrm{Sn}(\mathrm{OH})_6{ }^{2-}\) ऐसी होती हैं, जिनके केन्द्रीय परमाणु sp3d2 संकरित होते हैं।
प्रश्न 2.
TICl3 की तुलना में BCl3 के उच्च स्थायित्व को आप कैसे समझाएँगे ?
उत्तर:
बोरॉन (B) परमाणु की स्थिति में, अक्रिय युग्म प्रभाव नगण्य होता है। इसका अर्थ है कि इसके तीनों संयोजी इलेक्ट्रॉन (2s2px1) क्लोरीन परमाणुओं के साथ बन्ध बनाने के लिए उपलब्ध हैं। इसलिए BCl3 स्थायी होता है। यद्यपि थैलियम (Tl) की स्थिति में, संयोजी s-इलेक्ट्रॉन (6s2) अधिकतम अक्रिय युग्म प्रभाव अनुभव करते हैं। अतः केवल संयोजी p-इलेक्ट्रॉन (6p1) बंध के लिए उपलब्ध होते हैं। इन परिस्थितियों में TlCl अत्यधिक स्थायी होते हैं, जबकि TlCl3 अपेक्षाकृत बहुत कम स्थायी होता है।
निष्कर्ष रूप से स्पष्ट है कि TlCl3 की तुलना में BCl3 उच्च स्थायी होता है।
प्रश्न 3.
बोरॉन ट्राइफ्लुओराइड लूइस अम्ल के समान व्यवहार क्यों प्रदर्शित करता है ?
उत्तर:
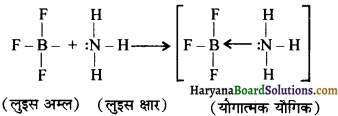
बोरॉन ट्राइफ्लुओराइड BF3 अणु में F परमाणु के इलेक्ट्रॉन-न्यून अणु हैं तथा यह स्थायी इलेक्ट्रॉनिक विन्यास प्राप्त करने के लिए एक इलेक्ट्रॉन युग्म ग्रहण करके लूइस अम्ल के समान व्यवहार प्रदर्शित करता है। उदाहरणार्थ-बोरॉन ट्राइफ्लुओराइड सरलतापूर्वक अमोनिया से एक एकांकी इलेक्ट्रॉन युग्म ग्रहण करके BF3.NH3 उपसहसंयोजक यौगिक बनाता है।
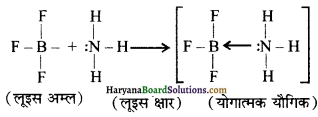
प्रश्न 4.
BCl3 तथा CCl4 यौगिकों का उदाहरण देते हुए जल के प्रति इनके व्यवहार के औचित्य को समझाइए।
उत्तर:
BCl3 में (B परमाणु sp2- संकंरित हैं), B परमाणु का अष्टक अपूर्ण है तथा इसका असंकरित 2p-कक्षक जल अणु से इलेक्ट्रॉन-युग्म ग्रहण करके योगात्मक उत्पाद बना सकता है।
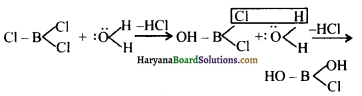
इस प्रकार जल से अभिक्रिया करने पर एक Cl परमाणु -OH समूह से प्रतिस्थापित हो जाता है। इसी प्रकार अन्य दो Cl परमाणु भो -OH समूह से प्रतिस्थापित हो जाते हैं।
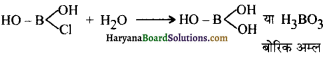
इससे प्रदर्शित होता है कि बोरॉन ट्राइक्लोराइड का जल-अपघटन हो जाता है, परन्तु यह CCl4 के साथ सम्भव नहीं है। कार्बन परमाणु का अष्टक पूर्ण होता है तथा H2O अणुओं के साथ योगात्मक उत्पाद बनने की कोई सम्भावना नहीं है। परिणामस्वरूप कार्बन टेट्राक्लोराइड जल-अपघटित नहीं होता। जल में मिलाने पर यह उसमें मिश्रित भी नहीं होता, अपितु एक पृथक् तैलीय पर्त बनाता है।

प्रश्न 5.
क्या बोरिक अम्ल प्रोटीनी अम्ल है? समझाइए।
उत्तर:
बोरिक अम्ल प्रोटीनी अम्ल नहीं है। यह एक लूइस अम्ल है तथा H2O अणु के हाइड्रॉक्सिल आयन से इलेक्ट्रॉन-युग्म ग्रहण करता है।
B(OH)3 + 2HOH → [B(OH)4]– + H3O+
प्रश्न 6.
क्या होता है, जब बोरिक अम्ल को गर्म किया जाता है?
उत्तर:
370K से अधिक ताप पर गर्म किए जाने पर बोरिक अम्ल (ऑर्थोबोरिक अम्ल) मेटाबोरिक अम्ल (HBO2) बनाता है, जो और अधिक गर्म करने पर बोरिक ऑक्साइड (B2O3) में परिवर्तित हो जाता है।

प्रश्न 7.
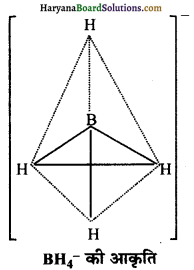
BF3 तथा BH4– की आकृति की व्याख्या कीजिए। इन स्पीशीज में बोरॉन के संकरण को निर्दिष्ट कीजिए।
उत्तर:
BF3 की आकृति- BF3 में sp2 संकरण होता है। जिस कारण इन कक्षकों की संख्या 3 होती है। तथा इन कक्षकों बीच 120° का कोण होता है जिससे इलेक्ट्रॉन युग्मों में पारस्परिक प्रतिकर्षण न्यूनतम रहता है तथा यह अणु त्रिकोणीय व समतल होता है।
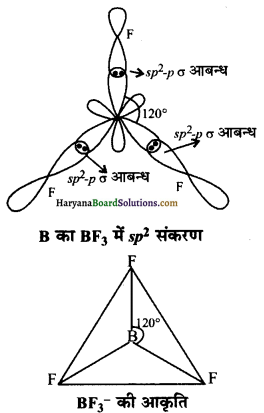
BH4– में B का संकरण sp3 होता है। इसकी आकृति चतुष्फलकीय होती है तथा 109°28′ का कोण होता है।
प्रश्न 8.
ऐलुमिनियम के उभयधर्मी व्यवहार दर्शाने वाली अभिक्रियाएँ दीजिए।
उत्तर:
ऐलुमिनियम अम्ल तथा क्षार दोनों से अभिक्रिया कर सकता है। इसलिए यह उभयधर्मी प्रकृति का होता है। उदाहरणार्थ-
2Al(s) + 6HCl(aq) → 2AlCl3(aq) + 3H2(g) तनु
2Al(s) + 2NaOH(aq) + 6H2O(l) → 2Na[Al(OH)4](aq) + 3H2(g)
प्रश्न 9.
इलेक्ट्रॉन न्यून यौगिक क्या होते हैं? क्या BCl3 तथा SiCl4 इलेक्ट्रॉन न्यून यौगिक हैं? समझाइए।
उत्तर:
इलेक्ट्रॉन न्यून यौगिक (Electron deficient Compounds)-वे यौगिक जिनके अणुओं में केन्द्रीय परमाणु एक या अधिक इलेक्ट्रॉन-युग्मों को ग्रहण करने की प्रवृत्ति रखते है, इलेक्ट्रॉन-न्यून यौगिक कहलाते हैं। इलेक्ट्रान-न्यून यौगिक को लूइस अम्ल भी कहा जाता है।
BCl3 तथा SiCl4 दाना इलक्ट्रान-न्यून योंगक है। B परमाणु मे रिक्त 2p कक्षक होते हैं, जबकि Si परमाणु में रिक्त 3-d कक्षक होते हैं। ये दोनों परमाणु इलेक्ट्रॉन-दाता स्पीशीज से इलेक्ट्रॉन युग्म ग्रहण कर सकते हैं। अत: BCl3 तथा SiCl4 दोनों इलेक्ट्रॉन-न्यून यौगिक हैं।
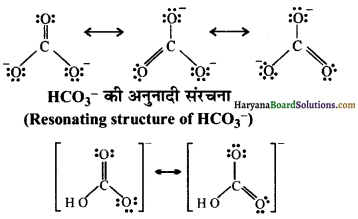
प्रश्न 10.
\(\mathrm{CO}_3^{2-}\) तथा \(\mathrm{HCO}_3^{-}\) की अनुनादी संरचनाएँ लिखिए।
उत्तर:
\(\mathrm{CO}_3^{2-}\) की अनुनादी संरचना (Resonating Structure of \(\mathrm{CO}_3^{2-}\) )
प्रश्न 11.
(क) \(\mathrm{CO}_3^{2-}\), (ख) हीरा तथा (ग) ग्रेफाइट में कार्बन की संकरण-अवस्था क्या होती है?
उत्तर:
(क) \(\mathrm{CO}_3^{2-}\) में कार्बन की संकरण-अवस्था sp2 होती है।
(ख) हीरे में कार्बन की संकरण-अवस्था sp3 होती है।
(ग) ग्रेफाइट में कार्बन की संकरण-अवस्था sp2 होती है।
प्रश्न 12.
संरचना के आधार पर हीरा तथा ग्रेफाइट के गुणों में निहित भिन्नता को समझाइए।
उत्तर:
हीरा तथा ग्रेफाइट में संरचनात्मक भिन्नता हीरा
- हीरे में क्रिस्टलीय जालक होता है। इसमें एक-दूसरे से बँधे कार्बन परमाणुओं का जाल होता है।
- प्रत्येक कार्बन परमाणु sp3 संकरित होता है तथा एकल सहसंयोजी बंध द्वारा चार अन्य कार्बन परमाणुओं से जुड़ा रहता है।
- प्रत्येक कार्बन परमाणु चतुष्फलक के केन्द्र पर स्थित होता है तथा अन्य चार कार्बन परमाणु चतुष्फलक के चारों कोनों पर स्थित होते हैं।
- C-C बंध की लम्बाई 154pm होती है। इसलिए हीरे में प्रबल सहसंयोजी बंधों का त्रिविमीय जाल होता है।
- यह अत्यन्त कठोर होता है। इसका गलनांक उच्च होता है।
ग्रेफाइट:
- ग्रेफाइट में परते 340pm की दूरी पर पृथक्कृत रहती हैं। इन परतो के बीच अत्यधिक दूरी यह प्रदर्शित करती है कि केवल दुर्बल वाण्डरवाल्स बल इन परतो को बाँधे रखते हैं।
- ग्रेफाइट में, प्रत्येक कार्बन परमाणु sp2 संकरण प्रदर्शित करता है तथा तीन अन्य कार्बन परमाणुओं से सहसंयोजी रूप से जुड़ा रहता है।
- प्रत्येक कार्बन परमाणु में चौथा इलेक्ट्रॉन π-बंध बनता है। अतः यह द्विविमीय षट्कोणीय वलय रखता है।
- वलय में C-C सहसंयोजी दूरी 142pm होती है जो प्रबल बंध को व्यक्त करती है। इन वलयों की व्यवस्था परते बनाती है।
- यह अत्यन्त कोमंल होता है। इसे मशीनों में शुष्क स्नेहक की भाँति प्रयोग किया जाता है।

प्रश्न 13.
निम्नलिखित कशनों को युक्तिसंगत कीजिए तथा रासायनिक समीकरण दीजिए-
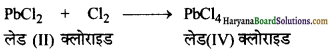
(क) लेड (II) क्लोराइड Cl2 से क्रिया करके PbCl4 देता है।
(ख) लेड (IV) क्लोराइड ऊष्मा के प्रति अत्यधिक अस्थायी है।
(ग) लेड एक आयोडाइड PbI4 नहीं बनाता है।
उत्तर:
(क) लेड (II) क्लोराइड Cl2 से क्रिया करके लेड (IV) क्लोराइड, (PbCl4) देता है क्योंकि क्लोरीन एक प्रबलतम ऑक्सीकारक है।
(ख) लेड IV ऑक्सीकरण अवस्था की तुलना में, II ऑक्सीकरण अवस्था में अधिक स्थायी होता है। इसलिए लेड (IV) क्लोराइड ऊष्मा के प्रति अत्यधिक अस्थायी होता है। यह गर्म करने पर विघटित होकर लेड (II) क्लोराइड बनाता है।
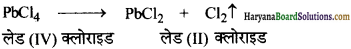
(ग) लेड एक आयोडाइड (PbI4) नहीं बनाता है; क्योंकि I– आयन के प्रबल अपचायक होने के कारण यह विलयन में Pb4+ आयन को Pb2+ आयन में अपचयित कर देता है।

प्रश्न 14.
BF3 में तथा \(\mathrm{BF}_4^{-}\) में बंध लम्बाई क्रमशः 130 pm तथा 143 pm होने के कारण बताइए।
उत्तर:
BF3 में तथा \(\mathrm{BF}_4^{-}\) में बोरॉन की संकरण-अवस्था निम्नलिखित प्रकार से दर्शाई जा सकती है-
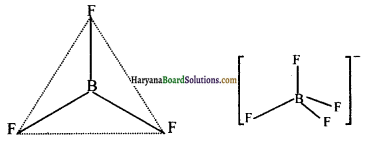
BF3 की आकृति समतलीय (Planer) है, इसमें sp2 संकरण पाया जाता है जबकि \(\mathrm{BF}_4^{-}\) की आकृति चतुष्फलकीय (tetrahedral) है, इसमें sp3 संकरण पाया जाता है, अतः दिए गए दोनों फ्लुओराइडों में बंध लम्बाइयों का अन्तर बोरॉन की संकरण-अवस्था में भिन्नता के कारण होता है।
प्रश्न 15.
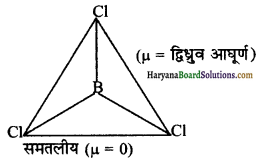
B-Cl आबन्ध द्विधुव आघूर्ण रखता है, किन्तु BCl3 अणु का द्विध्रुव आघूर्ण शून्य होता है। क्यों ?
उत्तर:
B-Cl आबन्ध एक निश्चित द्विध्रुव आघूर्ण रखता है; क्योंकि यह ध्रुवीय प्रकृति का होता है। परन्तु BCl3 अणु का द्विध्रुव आघूर्ण शून्य होता है; क्योंकि BCl3 अणु सममिताकार (समतलीय) होता है जिसमें आबन्ध ध्रुवणताएँ एक-दूसरे को निरस्त कर देती हैं।
प्रश्न 16.
निर्जलीय HF में ऐलुमिनियम ट्राइफ्लुओराइड अविलेय है, परन्तु NaF मिलाने पर घुल जाता है। गैसीय BF3 को प्रवाहित करने पर परिणामी विलयन में से ऐलुमिनियम ट्राइ फ्लुओराइड अवक्षेपित हो जाता है। इसका कारण बताइए।
उत्तर:
ऐलुमिनियम ट्राइफ्लुओराइड (AIF3) निर्जली HF में अविलेय होता है; क्योंकि इसकी प्रकृति सहसंयोजी होती है। यद्यपि यह NaF से अभिक्रिया करने पर एक संकुल यौगिक बनाता है, जो जल में विलेय होता है।
AlF3 + NaF → Na+[AlF4]–
(विलेय)
इस संकुल यौगिक को जलीय विलयन में BF3 की वाष्प बुलबुलों के रूप में प्रवाहित करने पर तोड़ा जा सकता है। परिणामस्वरूप ऐलुमिनियम ट्राइफ्लुओराइड पुनः अवक्षेपित हो जाता है।
Na+[AlF4]– + BF3→AlF3Na+[BF4]–↓
(अवक्षेप)
प्रश्न 17.
CO के विधैली होने का एक कारण बताइए।
उत्तर:
कार्बन मोनोक्साइड (CO) प्रकृति में पाई जाने वाली एक अत्यधिक विषैली गैस होती है। इसके विषैला होने का कारण यह है कि रक्त में उपस्थित हीमोग्लोबिन से संयुक्त होकर कार्बोक्सीहीमोग्लोबिन बना लेती है जो श्वसित की गई ऑक्सीजन को शरीर के विभिन्न भागों में ले जाने में असमर्थ होता है। इससे दम घुटने लगता है तथा अन्ततः मृत्यु हो जाती है।
प्रश्न 18.
CO2 की अधिक मात्रा भूमण्डलीय ताप वृद्धि के लिए उत्तरदायी कैसे है?
उत्तर:
CO2 में मेथेन के समान ऊष्मा अवशोषित करने की प्रवृत्ति होती है। इसे हरित-गृह गैस (green house gas) भी कहते हैं। इस प्रवृत्ति के कारण वायुमण्डल में इसकी अत्यधिक सान्द्रता भूमण्डलीय ताप वृद्धि के लिए उत्तरदायी होती है।
प्रश्न 19.
डाइबोरेन तथा बोरिक अम्ल की संरचना समझाइए।
उत्तर:
(क) डाइबोरेन की संरचना (Structure of Diborane)डाइबोरेन की संरचना को चित्र द्वारा दर्शाया गया है। इसमें सिरे वाले चार हाइड्रोजन परमाणु तथा दो बोरॉन परमाणु एक ही तल में होते हैं। इस तल के ऊपर तथा नीचे दो सेतु बंधित हाइड्रोजन परमाणु होते हैं। सिरे वाले चार B-H बंध सामान्य द्विकेन्द्रीय-द्विइलेक्ट्रॉन (two centretwo electron) बंध होते हैं तथा दो सेतु बन्ध (B-H-B) भिन्न प्रकार के होते हैं, जिन्हें ‘त्रिकेन्द्रीय-द्विइलेक्ट्रॉन बंध’ कहते हैं।
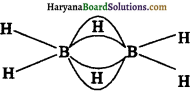
डाइबोरेन में प्रत्येक बोरॉन परामाणु sp3 संकरित होता है।
(ख) बोरिक अम्ल की संरचना (Structure of Boric acid)ठोस अवस्था में, बोरिक अम्ल की परतदार संरचना होती है, जहाँ समतलीय BO3 की इकाइयाँ हाइड्रोजन बंध द्वारा एक-दूसरे से 318pm की दूरी पर जुड़ी रहती हैं। बोरिक अम्ल में बोरॉन परमाणु sp2 संकरित होता है।
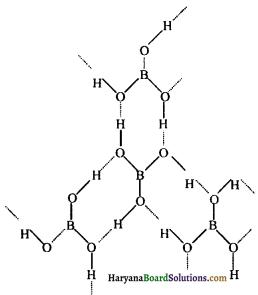
बोरिक अम्ल की संरचना में बिन्दुकृत रेखाएँ हाइड्रोजन आबन्ध को प्रदर्शित करती हैं।
प्रश्न 20.
क्या होता है, जब-
(क) बोरेक्स को अधिक गर्म किया जाता है।
(ख) बोरिक अम्ल को जल में मिलाया जाता है।
(ग) ऐल्युमिनियम की तनु NaOH से अभिक्रिया कराई जाती है।
(घ) BF3 की क्रिया अमोनिया से की जाती है।
उत्तर:
(क) जब बोरेक्स के चूर्ण को बुन्सन बर्नर की ज्वाला में अधिक गर्म किया जाता है, तो सर्वप्रथम यह जल के अणु का निष्कासन करके फूल जाता है। पुन: गर्म करने पर यह एक पारदर्शी द्रव में परिवर्तित हो जाता है, जो काँच के समान एक ठोस में परिवर्तित हो जाता है। इसे बोरेक्स मनका कहते हैं।
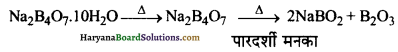
(ख) यह जल में घुल जाता है; क्योंकि यह इलेक्ट्रॉन-न्यून यौगिक है।
B(OH)3 + H – OH → [B(OH)4]– + H+
या
[H3BO3]
(ग) ऐलुमिनियम NaOH विलयन में घुलकर एक विलेय संकुल बनाता है तथा हाइड्रोजन गैस मुक्त करता है।
2Al(s) + 2NaOH(aq) + 6H2O(l) → 2Na+[Al(OH)4]–(aq) + 3H2(g)
(घ) BF3 (व्यवहार में लूइस अम्ल) NH3 (व्यवहार में लूइस-क्षारक) के साथ योगात्मक यौगिक बनाता है।

प्रश्न 21.
निम्नलिखित अभिक्रियाओं को समझाइए जब-
(क) कॉपर की उपस्थिति में उच्च ताप पर सिलिकन को मेथिल क्लोराइड के साथ गर्म किया जाता है।
(ख) सिलिकन डाइऑक्साइड की क्रिया ह्यइड्रोजन फ्लुओराइड के साथ की जाती है।
(ग) CO को ZnO के साथ गर्म किया जाता है।
(घ) जलीय ऐलुमिना की क्रिया जलीय NaOH के साथ की जाती है।
उत्तर:
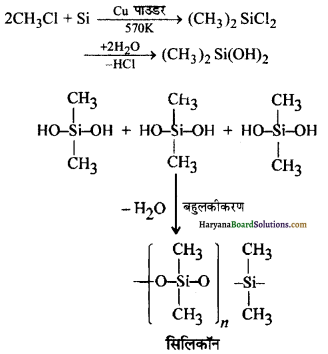
(क) कॉपर पाउडर (उत्प्रेरक) की उपस्थिति में उच्च ताप (570K) पर सिलिकन को मेथिल क्लोराइड के साथ गर्म करने पर डाइमेथिल डाइक्लोरोसिलेन प्राप्त होता है, जिसके जल-अपघटन के उपरान्त संघनन बहुलकीकरण द्वारा श्रृंखला बहुलक प्राप्त होते हैं।
(ख) सिलिकन डाइऑक्साइड की क्रिया हाइड्रोजन फ्लुओराइड के साथ होने पर सिलिकन टेट्राफ्लुओराइड (SiF4) बनता है।
SiO2 + 4HF → SiF4 + 2 H2O
(ग) CO, जो एक प्रबल अपचायक है, के द्वारा ZnO का अपचयन Zn में हो जाता है।
ZnO + CO → Zn + CO2
(घ) दोनों यौगिक दाब के अन्तर्गत गर्म किए जाने पर विलेय संकुल बनाते हैं।
Al2O3(s) + 2NaOH(aq) + 3H2O(l) → 2Na[Al(OH)4](aq)
प्रश्न 22.
कारण बताइए-
(क) सान्द्र HNO3 का परिवहन ऐलुमिनियम के पात्र द्वारा किया जा सकता है।
(ख) तनु NaOH तथा ऐलुमिनियम के टुकड़ों के मिश्रण का प्रयोग अपवाहिका खोलने के लिए किया जाता है।
(ग) ग्रेफाइट शुष्क स्नेहक के रूप में प्रयुक्त होता है।
(घ) हीरे का प्रयोग अपघर्षक के रूप में होता है।
(ङ) वायुयान बनाने में ऐलुमिनियम मिश्र धातु का प्रयोग होता है।
(च) जल को ऐलुमिनियम पात्र में पूरी रात नहीं रखना चाहिए।
(छ) संचरण केबल बनाने में ऐलुमिनियम तार का प्रयोग होता है।
उत्तर:
(क) सान्द्र HNO3 प्रारम्भ में ही ऐलुमिनियम से क्रिया करके ऐलुमिनियम ऑक्साइड (Al2O3) बना लेता है, जो पात्र के भीतर एक रक्षी-लेपन कर देता है। इस प्रकार धात्विक पात्र निष्क्रिय (passive) हो जाता है तथा फिर अम्ल से क्रिया नहीं करता। इसलिए अम्ल का परिवहन ऐलुमिनियम के पात्र द्वारा सुरक्षापूर्वक किया जा सकता है।
(ख) ऐलुमिनियम तनु NaOH में घुलकर H2 मुक्त करता है। यह हाइड्रोजन गैस अपवाहिका खोलने में सहायता करती है।
2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2↑
(ग) ग्रेफाइट में sp2- संकरित कार्बन होता है तथा इसकी परतीय संरचना होती है। व्यापक पृथक्करण तथा दुर्बल अन्तरपर्तीय बन्धों के कारण इसकी दो समीपवर्ती परते एक-दूसरे पर सरलतापूर्वक फिसल जाती हैं। इस कारण इसे शुष्क स्नेहक की भाँति उन मशीनों में प्रयुक्त किया जा सकता है जिनमें किसी कारणवश तैलीय स्नेहक प्रयुक्त न किए जा सकते हों।
(घ) हीरा समस्त ज्ञात पदार्थों में कठोरतम पदार्थ होता है। अत: इसका प्रयोग अपघर्षक (abrasive) तथा काँच काटने में किया जाता है।
(ङ) ऐलुमिनियम मिश्रधातु-मैग्नेलियम तथा ड्यूरैलियम जिनमें लगभग 95% धातु है, को वायुयान बनाने में प्रयोग किया जाता है। इस कारण ये हल्के, परन्तु मजबूत होते हैं। इसके अतिरिक्त इन पर जंग भी नहीं लगता है।
(च) जल को ऐलुमिनियम पात्र में पूरी रात नहीं रखना चाहिए; क्योंकि लम्बे समय तक नमी तथा ऑक्सीजन से धातु संक्षारित हो सकती है।
(छ) ऐलुमिनियम सामान्यतया वायु तथा नमी से प्रभावित नहीं होती तथा इसकी विद्युत-चालकता कॉपर से दोगुनी होती है। इसलिए संचरण केबल बनाने में ऐलुमिनियम तार का प्रयोग होता है।
प्रश्न 23.
कार्बन से सिलिकॉन तक आयनीकरण एन्थैल्पी में प्रघटनीय कमी होती है। क्यों ?
उत्तर:
कार्बन से सिलिकॉन तक आयनीकरण में प्रघटनीय कमी होती है; क्योंकि कार्बन की परमाणु त्रिज्या (77 pm) की तुलना में सिलिकॉन की परमाणु त्रिज्या अधिक (118 pm) होती है। इसलिए इलेक्ट्रॉनों का निष्कासन सरलतापूर्वक हो जाता है। सिलिकॉन से जर्मेनियम तक आयनन एन्थैल्पी में कमी प्रघटनीय नहीं होती; क्योंकि तत्वों के परमाणु आकार एकसमान रूप से बढ़ते हैं।
प्रश्न 24.
Al की तुलना में Ga की कम परमाण्वीय त्रिज्या को आप कैसे समझयेंगे ?
उत्तर:
Al की तुलना में Ga की कम परमाण्वीय त्रिज्या को प्रथम संक्रमण श्रेणी (Z = 21 से 30) के दस तत्वों की उपस्थिति के आधार पर समझाया जा सकता है। इनमें इलेक्ट्रॉन 3d-कक्षकों में होते हैं। चूँकि d-कक्षकों का आकार p-कक्षकों की तुलना में अधिक होता है; अतः अन्तरस्थ इलेक्ट्रॉनों के पास नाभिकीय आवेश में वृद्धि के प्रभाव को निरस्त करने के लिए पर्याप्त परिरक्षण प्रभाव नहीं होता। इसलिए Ga की स्थिति में प्रभावी नाभिकीय आवेश का मान कम होता है। इससे अपवादस्वरूप Ga का परमाणु आकार घट जाता है जिसे वास्तव में बढ़ा होना चाहिए था।
प्रश्न 25.
अपररूप क्या होता है? कार्बन के दो महत्त्वपूर्ण अपररूप हीरा तथा ग्रेफाइट की संरचना का चित्र बनाइए। इन दोनों अपररूपों के भौतिक गुणों पर संरचना का क्या प्रभाव पड़ता है ?
उत्तर:
प्रकृति में शुद्ध कार्बन दो रूपों में पाया जाता है—हीरा तथा ग्रेफाइट। यदि हीरे अथवा ग्रेफाइट को वायु में अत्यधिक गर्म किया जाए तो यह पूर्ण रूप से जल जाते हैं तथा कार्बन डाइऑक्साइड बनाते हैं। जब हीरे तथा ग्रेफाइट की समान मात्रा दहन की जाती है, तब कार्बन डाइऑक्साइड की बराबर मात्रा उत्पन्न होती है तथा कोई अवशेष नहीं बचता। इन तथ्यों से स्पष्ट है कि हीरा तथा ग्रेफाइट रासायनिक रूप से एक-समान हैं तथा केवल कार्बन परमाणुओं से बने हैं। इनके भौतिक गुण अत्यधिक भिन्न होते हैं। अतः इस प्रकार के गुणों को प्रदर्शित करने वाले तत्वों को अपररूप कहते हैं। हीरा तथा ग्रेफाइट कॉर्बन के दो प्रमुख क्रिस्टलीय अपररूप हैं।
1. हीरा – हीरा में क्रिस्टलीय जालक होता है। इसमें प्रत्येक परमाणु sp3 संकरित होता है तथा चतुष्फलकीय ज्यामिति से अन्य चार कार्बन परमाणुओं से जुड़ा रहता है। इसमें कार्बन-कार्बन बंध लम्बाई 154 pm होती है। कार्बन परमाणु दिक् (space) में दृढ़ त्रिविमीय जालक (rigid three dimensional network) का निर्माण करते हैं।
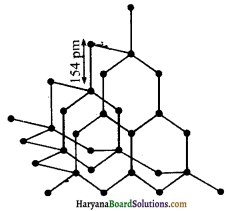
इस संरचना में सम्पूर्ण जालक में दिशात्मक सहसंयोजक बंध उपस्थित रहते हैं। इस प्रकार विस्तृत सहसंयोजक बन्धन को तोड़ना कठिन कार्य होता है। अतः हीरा पृथ्वी पर पाया जाने वाला सर्वाधिक कठोर पदार्थ है। इसका उपयोग धार तेज करने के लिए अपघर्षक (abrasive) के रूप में, डाई बनाने में तथा विद्युत-प्रकाश लैम्प में टंगस्टन तन्तु (filament) बनाने में होता है।
2. ग्रेफाइट – ग्रेफाइट की पर्त्त्र्य संरधना (layered structure) होती है। ये पर्तें वाण्डरवाल बल द्वारा जुड़ी रहती हैं।
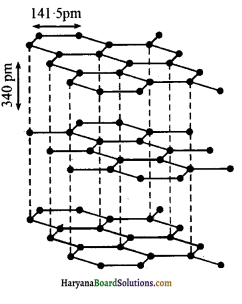
इस कारण ग्रेफाइट चिकना (slippery) तथा मुलायम (soft) होता है। दो पर्तों के मध्य की दूरी 340 pm होती है। प्रत्येक पर्त में कार्बन परमाणु षट्कोणीय वलय (hexagonal rings) के रूप में व्यवस्थित होते हैं जिसमें C-C बंध लम्बाई 141.5 pm होती है। षट्कोणीय वलय में प्रत्येक कार्बन परमाणु sp2 संकरित होता है। प्रत्येक कार्बन परमाणु तीन निकटवर्ती कार्बन परमाणुओं से तीन सिग्मा बंध बनाता है। इसका चौथा इलेक्ट्रॉन π-बंध बनाता है। सम्पूर्ण पर्त में इलेक्ट्रॉन विस्थानीकृत होते हैं। इलेक्ट्रॉन गतिशील होते हैं; अत: ग्रेफाइट विद्युत का सुचालक होता है। उच्च ताप पर जिन मशीनों में तेल का प्रयोग स्नेहक (lubricant) के रूप में नहीं हो सकता है, उनमें ग्रेफाइट शुष्क स्नेहक का कार्य करता है।
3. फुलरीन्स (Fullerenes) – एच. डब्ल्यू. क्रोटो, ई. स्मैले तथा आर. एफ. कर्ल ने सन् 1985 में कार्बन में एक अन्य अपररूप फुलरीन की खोज की। इसी खोज के कारण इन्हें सन् 1996 में नोबेल पुरस्कार प्राप्त हुआ।
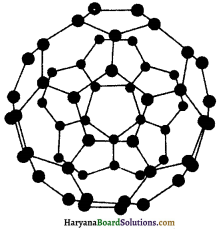
हीलियम, आर्गन आदि अक्रिय गैसों की उपस्थिति में जब ग्रेफाइट को विद्युत आर्क (electric arc) में गर्म किया जाता है, तब फुलरीन का निर्माण होता है। वाष्पित लघु Cn अणुओं को संघनित करने पर प्राप्त कज्जली पदार्थ (sooty material) में मुख्य रूप से C60 कुछ अंश C70 तथा अतिसूक्ष्म मात्रा में 350 या अधिक समसंख्या में कार्बन फुलरीन में पाए गए हैं। फुलरीन कार्बन का शुद्धतम रूप हैं; क्योंकि फुलरीन में किसी प्रकार का झूलता बंध (dangling bond) नहीं होता है। फुलरीन की संरचना पिंजरानुमा (cage-like) होती है। C60 अणु की आकृति सॉकर बॉल के समान होती है। इसे बकमिन्ट्टर फुलरीन (buckminstefulerene) कहते हैं।
इसमें छह सदस्यीय बीस वलय तथा पाँच सदस्यीय बारह वलय होती हैं। एक छह सदस्यीय वलय छह अथवा पाँच सदस्यीय वलय के साथ संगलित (fused) रहती है, जबकि पाँच सदस्यीय नलय केवल छ: सदस्यीय वलय के साथ संगलित अवस्था में रहती है। सभी कार्बन परमाणु समान होते हैं तथा sp2-संकरित होते हैं। प्रत्येक कार्बन परमाणु अन्य तीन कार्बन परमाणुओं के साथ तीन आबन्ध बनाता है।
चौथा इलेक्ट्रॉन पूरे अणु पर विस्थानीकृत रहता है, जो अणु को ऐरोमैटिक गुण प्रदान करता है। दस गेंदनुमा अणु में 60 उदग्र (vertices) होते हैं। प्रत्येक उदग्र पर एकल कार्बन परमाणु होता है। इस पर दोनों एकल तथा द्विबन्ध होते हैं, जिसकी C-C की लम्बाई क्रमशः 143.5 pm तथा 138.3 pmहोती है। गोलाकार फुलरीन को ‘बकी बॉल’ (Bucky ball) भी कहते हैं।
(4) अन्य अपररूप (Other Allotropes) – कार्बन तत्व के अन्य अपररूप भी होते हैं जैसे-कार्बन ब्लैक, कोक, चारकोल आदि। ये सभी अशुद्ध अपररूप कहलाते हैं।

प्रश्न 26.
(क) निम्नलिखित ऑक्साइड को उदासीन, अम्लीय, क्षारीय तथा उभयधर्मी ऑक्साइड के रूप में वर्गीकृत कीजिए-
CO, B2O3, SiO2, Al2O3, PbO2, TI2O3
(ख) इनकी प्रकृति को दर्शाने वाली रासायनिक अभिक्रिया लिखिए।
उत्तर:
(क) उदासीन ऑक्साइड : CO
अम्लीय ऑक्साइड : SiO2, CO2, B2O3
क्षारीय ऑक्साइड : Tl2O3, PbO2
उभयधर्मी ऑक्साइड : Al2O3
(ख) CO – उदासीन
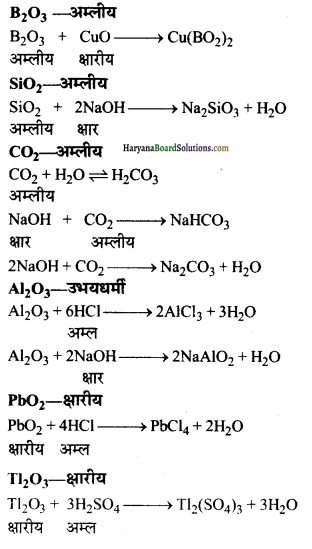
प्रश्न 27.
कुछ अभिक्रियाओं में थैलियम, ऐलुमिनियम से समानता दर्शाता है, जबकि अन्य में यह समूह-I के धातुओं से समानता दर्शाता है। इस तथ्य को कुछ प्रमाणों के द्वारा सिद्ध करें।
उत्तर:
थैलियम की ऐलुमिनियम से समानता
- दोनों का बाह्यतम इलेक्ट्रॉनिक विन्यास ns2np1 होता है।
- दोनों वायु में ऑक्साइड बनने के कारण धूमिल पड़ जाते हैं।
- Al तथा Tl दोनों के फ्लोराइड आयनिक होते हैं तथा इनका गलनांक उच्च होता है।
- Al तथा Tl दोनों ही +3 ऑक्सीकरण संख्या भी दर्शाते हैं।
थैलियम की समूह -I की धातुओं से समानता
- थैलियम एवं समूह -I दोनों ही +1 ऑक्सीकरण संख्या दर्शाते हैं।
- NaOH के समान Tl(OH) जल में विलेय होकर प्रबल क्षारीय विलयन बनाता है।
- क्षार धातुओं के समान, थैलियम ऐलुमिनियम लवणों के साथ द्विक लवण बनाता है।

प्रश्न 28.
जब धातु X की क्रिया सोडियम हाइड्रु क्साइड के साथ की जाती है तो श्वेत अवक्षेप (A) प्राप्त होता है, जो NaOH के आधिक्य में विलेय होकर विलेय संकुल (B) बनाता है। यौगिक (A) तनु HCl में घुलकर (C) बनाता है। यौगिक(A) को अधिक गर्म किए जाने पर यौगिक (D) बनता है, जो एक निष्कर्षित घातु के रूप में प्रयुक्त होता है। X,A,B, C तथा D को पहचानिए तथा इनकी पहचान के समर्थन में उपयुक्त समीकरण दीजिए।
उत्तर:
दी गई परिस्थितियों के अनुसार धातु X ऐलुमिनियम है। वे अभिक्रियाएँ, जिनमें ऐलुमिनियम भाग लेकर यौगिक A,B,C तथा D बनाता है, अग्रलिखित हैं-
(i) ऐलुमिनियम (X) को NaOH के साथ गर्म करने पर यह Al(OH)3 का सफेद् अवक्षेप बनाता है अर्थात् यौगिक (A) बनाता है जो NaOH के आधिक्य में घुलकर विलेय संकर (B) बनाता है।
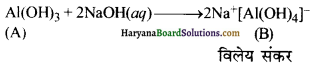
(ii) यौगिक (A) तनु HCl में घुलकर ऐलुमिनियम क्लोराइड (C) बनाता है।

(iii) गर्म करने पर Al(OH)3, ऐलुमिना (D) में परिवर्तित हो जाता है।
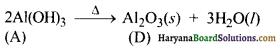
Al2O3 का प्रयोग ऐलुमिनियम के निष्कर्षण में किया जाता है।
प्रश्न 29.
निम्नलिखित से आप क्या समझते हैं?
(क) अक्रिय युग्म प्रभाव,
(ख) अपररूप,
(ग) श्रृंखलन।
उत्तर:
(क) अक्रिय युग्म प्रभाव (Inert pair effect) – इलेक्ट्रॉनिक कोश विन्यास, (n – 1)d10 ns2np1 वाले तत्व में, d-कक्षक के इलेक्ट्रॉन दुर्बल परिरक्षण प्रभाव (poor shielding effect) प्रदर्शित करते हैं। इसलिए ns2 इलेक्ट्रॉन नाभिक के धनावेश द्वारा अधिक दृढ़ता से बँधे रहते हैं। इस प्रबल आकर्षण के परिणामस्वरूप, ns2 इलेक्ट्रॉन युग्मित रहते हैं तथा बन्ध में भाग नहीं लेते हैं अर्थात् अक्रिय रहते हैं। यह प्रभाव अक्रिय युग्म प्रभाव कहलाता है। इस स्थिति में, ns2np1 विन्यास में, तीन इलेक्ट्रॉनों में से केवल एक इलेक्ट्रॉन बंध-निर्माण में भाग लेता है।
(ख) अपररूप (Allotropes) – किसी तत्व का समान रासायनिक अवस्था में दो या अधिक भिन्न-रूपों में पाया जाना अपररूपता कहलाता है। तत्व के ये विभिन्न रूप अपररूप कहलाते हैं। किसी तत्व के सभी अपररूपों के रासायनिक गुण समान होते हैं, परन्तु इनके भौतिक गुणों मे अन्तर होता है।
(ग) शृंखलन (Catenation) – कार्बन में अन्य परमाणुओं के साथ सहसंयोजक बंध द्वारा जुड़कर लम्बी शृंखला या.वलय बनाने की प्रवृत्ति होती है। इस प्रवृत्ति को श्रृंखलन कहते हैं। C-C बंध अधिक प्रबल होने के कारण ऐसा होता है।
प्रश्न 30.
एक लवण X निम्नलिखित परिणाम देता है-
(क) इसका जलीय विलयन लिटमस के प्रति क्षारीय होता है।
(ख) तीव्र गर्म किए जाने पर यह काँच के समान ठ्रेस में स्वेदित हो जाता है।
(ग) जब X के गर्म विलयन में सान्द्र H2SO4 मिलाया जाता है तो एक अम्ल Z का श्वेत क्रिस्टल बनता है।
उपर्युक्त अभिक्रियाओं के समीकरण लिखिए और X, Y तथा Z को पहचानिए।
उत्तर:
दिए गए परिणामों से स्पष्ट है कि लवण X बोरेक्स (Na2B4O7) है।
(क) बोरेक्स का जलीय विलयन क्षारीय प्रकृति का होता है तथा लाल लिटमस को नीला कर देता है।

(ख) बोरेक्स तीव्र गर्म किए जाने पर स्वेदित (swell) हो जाता है तथा क्रिस्टलन जल के अणु खोकर ठोस (Y) बनाता है।
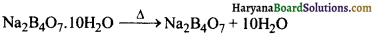
(ग) सान्द्र H2SO4 से अभिक्रिया करने पर, बोरेक्स बोरिक अम्ल (H3BO3) बनाता है। जब इसे विलयन से क्रिस्टलीकृत किया जाता है तो श्वेत क्रिस्टलों (Z) के रूप में होता है।
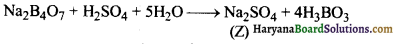
प्रश्न 31.
सन्तुलित समीकरण दीजिए-
(क) BF3 + LiH →
(ख) B2H6 + H2O →
(ग) NaH + B2H6 →
(घ) H3BO3
(ङ) Al + NaOH →
(च) B2H6 + NH3 →
उत्तर:

प्रश्न 32.
CO तथा CO प्रत्येक के संश्लेषण के लिए एक प्रयोगशाला तथा एक औद्योगिक विधि दीजिए।
उत्तर:
(क) कार्बनमोनोऑक्साइड (Carbon Mono oxide)
प्रयोगशाला विधि (Laboratory Method)-सान्द्र सल्फ्यूरिक अम्ल का 373K पर फॉर्मिक अम्ल के द्वारा निर्जलीकरण कराने पर अल्प मात्रा में शुद्ध कार्बन मोनो ऑक्साइड प्राप्त होती है।
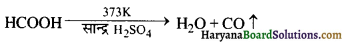
औद्योगिक विधि (Industrial Method)-औद्योगिक रूप में इसे कोक पर भाप (Steam) प्रवाहित करके बनाया जाता है। इस प्रकार CO तथा H2 का प्राप्त मिश्रण ‘वाटर गैस’ अथवा ‘संश्लेषण गैस’ (synthesis gas) कहलाता है।
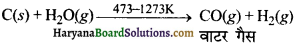
जब भाप के स्थान पर वायु का प्रयोग किया जाता है, तब CO तथा N2 का मिश्रण प्राप्त होता है। इसे प्रोड्यूसर गैस कहते हैं।
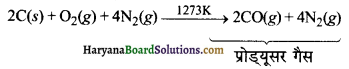
(ख) कार्बन डाइऑक्साइड (Carbon dioxide)
प्रयोगशाला विधि (Laboratory Method) – प्रयोगशाला में इसे कैल्सियम कार्बोनेट पर तनु HCl की अभिक्रिया द्वारा बनाया जाता है।
CaCO3(s) + 2HCl(aq) → CaCl2(aq)+CO2(g)+H2O(l)
कैल्सियम – कैल्सियम कार्बन डाइ
कार्बोनेट – क्लोराइड ऑक्साइड
औद्योगिक विधि (Industrial Method) – औद्योगिक रूप में चूना पत्थर (lime-stone) को गर्म करके CO2 बनाई जा सकती है।

प्रश्न 33.
बोरेक्स के जलीय विलयन की प्रकृति कौन-सी होती है?
(क) उदासीन
(ख) उभयधर्मी
(ग) क्षारीय
(घ) अम्लीय।
उत्तर:
(ग) क्षारीय।
प्रश्न 34.
बोरिक अम्ल के बहुलकीय होने का कारण ?
(क) इसकी अम्लीय प्रकृति है
(ख) इसमें हाइड्रोजन बंधों की उपस्थिति है
(ग) इसकी एकक्षारीय प्रकृति है
(घ) इसकी ज्यामिति है।
उत्तर:
(ख) इसमें हाइड्रोजन बंधों की उपस्थिति है।
प्रश्न 35.
डाइबोरेन में बोरॉन का संकरण कौन-सा होता है?
(क) sp
(ख) sp2
(ग) sp3
(घ) dsp2
उत्तर:
(ग) sp3
प्रश्न 36.
ऊष्मागतिकीय रूप से कार्बन का सर्वाधिक स्थायी रूप कौन-सा है?
(क) हीरा
(ख) ग्रेफाइट
(ग) फुलरीन्स
(घ) कोयला।
उत्तर:
(ख) ग्रेफाइट।
प्रश्न 37.
निम्नलिखित में से समूह-14 के तत्वों के लिए कौन-सा कथन सत्य है ?
(क) +4 ऑक्सीकरण अवस्था प्रदर्शित करते हैं।
(ख) +2 तथा +4 ऑक्सीकरण अवस्था प्रदर्शित करते हैं।
(ग) M2- तथा M4+ आयन बनाते हैं।
(घ) M2+ तथा M4- आयन बनाते हैं।
उत्तर:
(ख) +2 तथा +4 ऑक्सीकरण अवस्था प्रदर्शित करते हैं।
प्रश्न 38.
यदि सिलिकॉन-निर्माण में प्रारमिभक पदार्थ RSiCl3 है तो बनने वाले उत्पाद की संरचना बताइए।
उत्तर:
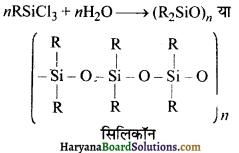
![]()
![]()
![]()
![]()
![]()