Haryana State Board HBSE 7th Class Science Solutions Chapter 5 Acids, Bases and Salts Textbook Exercise Questions and Answers.
Haryana Board 7th Class Science Solutions Chapter 5 Acids, Bases and Salts
HBSE 7th Class Science Acids, Bases and Salts Textbook Questions and Answers
Question 1.
State differences between acids and bases.
Answer:
| Acids | Bases |
| 1. A substance which dissolves in water to give hydrogen ions as the only positively charged ions is called an acid. | 1. A substance which react with acids to form salt and water as only products is called a base. |
| 2. They have a sour taste. | 2. These have a better taste. |
| 3. They turn blue litmus red. | 3. Bases turn red litmus blue. |
Question 2.
Ammonia is found is many household products, such as window cleaners. It turns red litmus blue, what is its nature?
Answer:
Basic.
![]()
Question 3.
Name the source from which litmus solution is obtained. What is the use of this solution?
Answer:
Litmus solution is extracted from lichens. It has a mauve (purple) colour in distilled water. When added to an acidic solution, it turns red and when added to a basic solution, it turns blue. It is available in the form of a solution, or in the form of strips of paper.
Question 4.
Is the distilled water acidic/basic/ neutral? How would you verify it?
Answer:
The distilled water is neutral. Put a drop of the distilled water on a strip of the red litmus paper with the help of a droper. We observe that colour of the litmus paper as not change.
The solutions which do not change the colour of either red or blue litmus are known as neutral solutions. These substances are neither acidic nor basic.
Question 5.
Describe the proeess of neutralization with the help of an example.
Answer:
The process due to! which an acid completely reacts with a base to form salt and water as the only products is called neutralization.
Acid + Base → Salt + Water
Example: Hydrochloric add (HCl) + Sodium hydroxide (NaOH)
Sodium chloride (NaCl) + Water (H2O)
Question 6.
Make “T” if the statement is true and ‘F’ if it is false.
(i) Nitric acid turn red litmus blue.
(ii) Sodium hydroxide turns blue litmus red.
(iii) Sodium hydroxide and hydrochloric acid neutralise each other and form salt and water.
(iv) Indicator is a substance which shows different colours in acidic and basic solutions.
(v) Tooth decay is caused by the presence of a base.
Answer:
(i) Flase
(ii) Flase
(iii) True
(iv) True
(v) True.
Question 7.
Dorji has a few bottles of soft drink in his restaurant. But unfortunately, these are not labelled. He has to serve the drinks on the demand of customers. One customer wants acidic drink, another wants basic and third one wants neutral drink; How will Dorji decide which drink is to be served to whom?
Answer:
Do yourself.
Question 8.
Explain why?
(a) An antacid tablet is taken when you suffer from acidity.
(b) Calamane solution is applied on the skin when an ant bites.
(c) Factory waste is neutralised before disposing it into the water bodies.
Answer:
(a) Our stomach contains hydrochloric acid. It help us to digest food. But too much of acid in the stomach causes indigestion. Sometimes indigestion is painful. To relieve indigestion, we take an antacid such as milk of magnesia, which contains magnesium hydroxide. It neutralises the effect of excessive acid.
(b) The sting of an ant contains formic acid. When an ant bites, it injects the acidic liquid into the skin. The effect of the sting can be neutralised by rubbing moist baking soda (sodium hydrogen carbonate) or calamine solution, which contains Zinc carbonate.
(c) The wastes of many factories contain acids. If they are allowed to flow into the water bodies, the acids will kill fish and other organisms. The factory wastes are, therefore, neutralised by adding basic substances.
Question 9.
Three liquids are given to you. One is hydrochloric acid, another is sodium hydroxide and third is a sugar solution. How will you Identify them? You have only turmeric indicator.
Answer:
Do yourself.
Question 10.
Blue litmus paper is dipped in a solution. It remains blue. What is the nature of the solution? Explain.
Answer:
The nature of the solution is neutral, because the solutions which do not change the colour of either red or blue litmus are known as neutral solution. These substances are neither acidic nor basic.
Question 11.
Consider the following statements:
(a) Acids and bases both change colour of all indicators.
(b) If an indicator gives a colour change with an acid, it does not give a change with a base.
(c) If an indicator changes colour with a base, it does not change colour with an acid.
(d) Change of colour in an acid and a base depends on the type of the indicator. Which of these statements are correct?
(i) All four
(ii) a and b
(iii) b and c
(iv) only d
Answer:
(ii) a and b.
![]()
Extended Learning – Activities And Projects
Question 1.
Using the knowledge of acids and bases, write a secret message with the help of baking soda and beet’root. Explain how it works.
(Hint: Prepare baking soda solution in water. Use this solution to write the message on a sheet of white paper with a cotton bud. Rub a slice of fresh beet root over the message.)
Answer:
Do yourself.
Question 2.
Prepare red cabbage juice by boiling a piece of red cabbage in water. Use it as an indicator and test the acidic and’ basic solutions with it. Present your observations in the form of a Table.
Answer:
Do yourself.
Question 3.
Bring the soil sample of your area, find out if it is acidic, basic or neutral. Discuss with farmers if they treat the soil in any manner.
Answer:
Do yourself.
Question 4.
Visit a doctor. Find out the medicines be prescribes to treat acidity. Ask him how acidity can be prevented.
Answer:
Do yourself.
![]()
HBSE 7th Class Science Acids, Bases and Salts Important Questions and Answers
Very Short Answer Type Questions
Question 1.
What is an acid Id
Answer:
A substance which dissolves in water to give hydrogen ions as the only positively charged ions’fs called an acid.
Question 2.
What are mineral acids? Give, three examples.
Answer:
The acids which are prepared from the minerals of the Earth are called mineral acids.
Examples: Sulphuric acid, Nitric acid, Hydrochloric acid.
Question 3.
What are organic acids? Give three examples.
Answer:
Organic acids are found in plant and animal products, for examples, oxalic acids, citric acid, acetic acid, etc.
Question 4.
A solution turns red litmus solution blue. Is the solution acidic or basic in nature?
Answer:
Basic.
Question 5.
Give two examples each of (i) Strong bases (ii) Weak bases.
Answer:
(i) Strong bases: Sodium hydroxide, potassium hydroxide.
(ii) Weak bases: Copper hydroxide, zinc hydroxide. .
Question 6.
What do you understand by the term “neutral substance?”
Answer:
The substances which do not bring about any change in the colour of common indicators are called neutral substances.
Question 7.
Name the acids present in:
(i) Vinegar
(ii) Lemon juice
Answer:
(i) Acetic acid (CH3COOH)
(ii) Citric acid.
![]()
Question 8.
Write the names of three mineral acids.
Answer:
(i) Sulphuric acid (H2SO4)
(ii) Nitric acid (HNO3) and
(iii)Hydrochloric acid (HCl) are mineral acids.
Question 9.
Which of the following are oxides?
H2O, CO, Co, NO, CO2 , NaOH
Answer:
Oxides: H2O, CO, NO and CO2.
Question 10.
From the following list select metallic oxide and non-metallic oxides: CaO, Fe2O3, N2O5, CO2, Na2O, SO2.
Answer:
(i) Metallic oxide: CaO, Fe2O3 and Na2O.
(ii) Non-metallic oxide: N2O5, CO2 and SO2.
Question 11.
What are indicators?
Answer:
The complex, naturally occurring substances, which change their colour on coming in contact with other substances are called indicators. Turmeric, litmus, china rose etc. are some indicators.
Question 12.
Name two neutral liquids.
Answer:
(i) Alcohol
(ii) Ether.
Question 13.
Name two neutral gases.
Answer:
(i) Hydrogen, (ii) Oxygen.
Question 14.
Name two neutral solids.
Answer:
(i) Common Salt
(ii) Sugar.
Question 15.
What do you understand by term concentrated acid?
Answer:
The acids which contain very little or no amount of water are called concentrated acids.
![]()
Question 16.
What are dilute acids?
Answer:
The acids which contain a large amount of water (or solutions of acids in water) are called dilute acids.
Question 17.
Name the kind of substance formed when an acid reacts with a base.
Answer:
Neutralised.
Question 18.
What is missing in the following equation?
Fat + ……. → Soap + Glycerol
Answer:
Fat + NaOH → Soap + Glyceral
Thus NaOH missing from the equation.
Question 19.
An element combines with oxygen to form an oxide. This oxide dissolves in water. This aqueous solution changes blue litmus to red. Write:
(i) The nature of element (metal or non-metal)
(ii) The general name of the oxide.
Answer:
(i) The element is non-metal.
(ii) General name of the oxide non-metallic oxide.
Question 20.
What happens when a non-metal oxide is dissolved in water?
Answer:
When a non-metal oxide is dissolved in water, it forms acids. Examples:
(i) CO2 + HO2 → H2CO3 (Carbonic acid).
(ii) SO2 + HO2 → H2SO3(Sulphuric acid).
Question 21.
State one characteristics, property of all acids.
Answer:
All acids contain replaceable hydrogen.
Question 22.
Name the gas obtained when a metal carbonate reacts with an acid.
Answer:
When a metal carbonate reacts with an acid, carbon dioxide gas is envolved. Example:
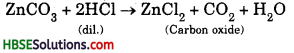
Question 23.
Write equations for the action of water on:
(i) MgO
(ii) Calcium oxide.
Answer:
(i) MgO + H2O → Mg(OH)2 (Magnesium hydroxide)
(ii) CaO + H2O → Ca(OH)2(Calcium hydroxide)
Question 24.
What are mineral acids? Give two examples.
Answer:
Mineral acids are those acids which are obtained from the earth. Examples, Sulphuric acid, Nitric acid.
Question 25.
Name the three salts which are commonly lised at home..
Answer:
Three, salts commonly used at home are:
(i) Sodium chloride
(ii)Sodium carbonate
(iii) Sodium hydrogen carbonate.
![]()
Question 26.
Give the chemical formula of slaked lime
Answer:
Chemical formula of slaked lime is Ca(OH)2
Question 27.
Name one/compound of calcium which is used for bleaching cloth.
Answer:
Bleaching powder.
Question 28.
What is the formula of sulphuric acid?
Answer:
The formula of sulphuric acid is H2SO4.
Question 29.
A solution turns red litmus paper blue. What does this indicate about the chemical nature of the solution?
Answer:
The chemical nature of the solution is acidic.
Question 30.
What do you understand by anhydrous salt?
Answer:
Hydrated salts loose their water of crystallisation upon heating. They love their shape and colour also and becomes powered. They are called anhydrous salts.
Question 31.
Name two normal salts.
Answer:
(i) Sodium Chloride (NaCl)
(ii) Potassium nitrate (KNO3).
Question 32.
Name two basic sdlts.
Answer:
(i) Calcium hydrdgen chloride [Ca(OH)Cl]
(ii) Magnesium hydroxy chloride [Mg(OH)Cl] etc.
Question 33.
Write the balanced chemical equations for the reaction that takes place when zinc, magnesium and sodium react with dilute hydrochloric acid.
Answer:
Reaction of hydrochloric acid (dil.) with Zn, Mg, and Na:
(i) Zn + 2HCl → ZnCl2 + H2
(ii) Mg + 2HCl → MgCl2 + H2
(iii) 2Na + 2HCl → 2NaCl + H2
Question 34.
Write two uses of slaked lime.
Answer:
Uses of slaked lime:
(i) It is used in manufacture of bleaching powder.
(ii) It is used as an important building materials
Question 35.
Complete the following equations:
(i) CO2 + H2O → ……………..
(ii) CO2 + 2Na0H → …………….. + ……………..
(iii) SO2 + H2O → ……………..
(iv) MgO + H2SO4 → …………….. + ……………..
(v) CaC03 + 2HCl → …………….. + ……………..
(vi) Na2O + H2O → ……………..
(vii) 2NaOH + H2SO4 → …………….. + ……………..
(viii) NaOH + HNO3 → …………….. + ……………..
(ix) CaO + H2SO4 → …………….. + ……………..
(x) MgO + 2HCl → ……………..
Answer:
(i) CO2 + H2O → H2CO3 (carbonic acid)
(ii) CO2 + 2Na0H → Na2CO3 + H2O (sodium carbonate)
(iii) SO2 + H2O → H2SO3 (suiphurous acid)
(iv) MgO + H2SO4 → MgSO4 + H2O (magnesium sulphate)
(v) CaC03 + 2HCl → CaCl2 + H2O + CO2 (Calcium Chloride)
(vi) Na2O + H2O → 2Na0H (sodium hydroxide)
(vii) 2NaOH + H2SO4 → Na2SO4 + H2O (sodium suphate)
(viii) NaOH + HNO3 → NaNO3 + H2O (sodiùm nitrate)
(ix) CaO + H2SO4 → CaSO4 + H2O (calcium sulphate)
(x) MgO + 2HCl → MgCl2 +H2O
![]()
Short Answer Type Questions
Question 1.
How can you test the presence of an acid in any substance?
Answer:
Acid can be tested in any substance by the following methods:
(i) Take a drop of the dilute solution of the substance on the tips of your tongue. If tastes sour, it is acidic.
(ii) Dip a blue litmus paper in the solution. If ‘ the colour of the litmus paper turns red, the solution is acidic.
Question 2.
What are organic acids? Give two examples.
Answer:
Soured milk, Lemon, Green mangoes and Karvanda are sour testing. All of them contain some naturally occuring acids. Such acids are also found in humans and plants materials. They are known as organic acids. The acids such as lactic acid, citric acid, tartaric acid and acetic acid are naturally occuring and known as organic acid.
Question 3.
What are the uses of mineral acids in industry?
Answer:
Use of’mineral acids in industry:
(i) Concentration sulphuric acid, nitric acid and hydrochloric acid are used in a large number of metallurgical operations, manufacture of fertilizers and preparations of large number of industrial chemicals.
(ii) Hydrochloric acid is used for removing the deposits from inside the boilers. This process is known as de-scaling.
Question 4.
What are indicators? Name two acid-base indicators.
Answer:
The substances which are used to identify whether a substance is an acid or base is called indicators. For example, litmus solutions give red colour with acids and blue colour with bases. Therefore, litmus is used as indicators. Substances which give different colours with acid and base are called acid-base indicators, for examples. Methyl orange, phenolphthalein.
Question 5.
What are acid salts? Give two examples.
Answer:
The salts which dissolves water to produce a solution, which behaves like an acid is called an acid salt.
Examples: Sodium hydrogen carbonate (NaHCO3), Potassium hydorgen carbonate (KHCO3), Sodium hydrogen sulphate etc.
Question 6.
How is potash alum prepared?
Answer:
A mixture of potassium sulphate and aluminium sulphate is dissolved in water and then the solution is concentrated. Potash alum is formed. It separates out as crystals.
Question 7.
Why are sodium bicarbonate and lemon juice used during indigestion?
Answer:
Sodium bicarbonate neutralizes the acidity in the stomach. Hence, it is used during indigestion. Lemon contains acid. It reacts with undigested food and softens it. Thus, lemon is also used in indigestion.
Question 8.
Why are brass vessels coated with tin or kalai?
Answer:
When brass vessels are used to cook food with tamarind or lemon juice, the acids present in it reacts with brass metal and corrode it. The kalai layer protects this vessels from the action of acids. Due to this reason brass vessels are coated with tin.
![]()
Question 9.
What is salt? Name any salts and give thier formulae.
Answer:
A substance formed by the neutralisation of an acid with a base is called salt.
Examples:
(i) Potassium nitrate (KNO3)
(ii) Sodiun: nitrate (NaNO3)
(iii) Calcium carbonate (CaCO3)
(iv) Zinc sulphate (ZnSO4)
(v) Copper sulphate (CuSO4).
Question 10.
Define an acid. Give three properties of acids.
Answer:
An acid is a substance which when dissolved in water gives hydrogen ions. Examples: H2SO4, HNO3 and HCl etc.
Properties of acids:
(i) They have a sour taste
(ii) They turn blue litmus solution red.
(iii) They have replaceable hydrogen ion.
Question 11.
What are bases? Mention three properties of alkalies.
Answer:
The substance which react with acids to form salt and water only are called bases. Examples: Cu(OH)2, Fe(OH)3, Pb(OH)2 etc.
Properties of alkalies:
(i) They tarn red litmus paper blue
(ii) They have a better taste.
(iii) They turn methyl orange from pink to yellow.
Question 12.
What are the uses of sodium bicarbonate?
Answer:
Uses of sodium bicarbonate:
(i) It is used in medicines to neutralise the acidity in the stomach.
(ii) It is used as backing power.
(iii) It is used in fire extinguisher.
Question 13.
What are the uses of common salt?
Answer:
Uses of common salt:
(i) It is an essential constituents of our diet.
(ii) It is used in the manufacture of soap.
(iii) It is used for glazing pottery. .
(iv) It is a starting material for the manufacture of chlorine, hydrochloric acid, washing soda and sodium hydroxide.
(v) It is used as food preservatives.
(vi) It turns freezing mixture with ice.
Question 14.
Give five uses of potassium nitrate.
Answer:
Uses of potassium nitrate:
(i) It is used in the manufacture of gun powder.
(ii) It is used in making fire works.
(iii) It is used in refrigeration.
(iv) It is used as fertilizers.
(v) It is used in glass industry.
Question 15.
Which acids is called ‘the king of the acids’? Give its uses.
Answer:
Sulphuric acid (H2SO4) is called “the king of the acids”.
Uses of sulphuric acid:
(i) It is used in the manufacture of detergents.
(ii) It is used as a dehydrating agent.
(iii) It is used in storage batteries.
(iv) It is used in petrol refining.
(v) It is used in textile, paper and leather industry.
(vi) It is used in fertilizers industry for the manufacture of ammonium sulphate and superphosphate of lime.
![]()
Question 16.
Explain with the help of chemical equations how the following acids are formed?
Carbonic acids, Sulphurous acid, Phosphoric acid.
Answer:
(i) Carbonic acid is prepared by dissolving carbon dioxide in water.
CO2 + H2O → H2CO3 (Carbonic acid)
(ii) Sulphurous acid is formed by dissolving sulphur dioxide in water.
SO2 + H2O → H2SO3 (Sulphurous acid)
(iii) Phosphoric acid is prepared by dissolving phosphorus pentaoxide in hot water.
P2O5 + 3H20 → 2H3PO4 (Phosphoric acid)
Acids, Bases and Salts Class 7 HBSE Notes
1. Curd, lemon juice, orange juice and vinegar taste sour. These substances taste sour because they contain acids. The chemical nature of such substances is acidic. The word acid comes from the Latin word acere which means sour. The acids in these substances are natural acids.
2. A substance which produces hydrogen (H+) ions when dissolved in water is called an acid. This definition was given by Arrhenius, a Swedish Chemist in 1912.
3. The acids which are prepared from the minerals of the Earth are called mineral acids.
Examples of Mineral Acids
| Name of Acid | Formula |
| Sulphuric acid | H2SO4 |
| Nitric acid | HNO4 |
| Hydrochloric acid | HCl |
| Phosphoric acid | H3PO4 |
4. The acids which dissolve in water to give a large number of positively charged hydrogen ions are called strong acids.
Examples of Strong Acids : Sulphuric acid, nitric acid, hydrochloric acid and phosphoric acid are examples of strong acids.
5. The acids which dissolve in water to give a small number of positively charged hydrogen ions are called weak acids.
Examples of Weak Acids : Carbonic acid, acetic acid, sulphurous acid, formic acid, etc., are examples of weak acids. In fact, most of the organic acid are weak acids.
6. The acids which contain very little or no amount of water are called concentrated acids.
7. The acids which contain a large amount of water (or solutions of acids in water) are called dilute acids.
8. General Physical Properties of Acids
(i) They have a sour taste.
(ii) They turn blue litmus solution red.
(iii) They turn methyl orange solution pink.
(iv) They do not affect phenolphthalein solution.
(v) Strong acids have a corrosive action on skin. They cause painful blisters.
(vi) Most of the acids are soluble in water.
(vii) Acids solutions are conductors of electricity.
9. Uses of Acids
| Acids | Uses |
| (i) Sulphuric acid | • manufacturing fertilizers, drugs, detergents, paints, plastic and artificial silk. • in strong batteries. • in paper, textile and leather industries. • as dehydrating agent. |
| (ii) Hydrochloric acid | • purifying metals and common selt. • making glucose, glue • bleaching textiles |
| (iii) Nitric acid | • extracting metals from ores brass, etc. • making designs on copper, • manufacturing explosives, fertilizers, perfumes, medicines and plastic. |
| (iv) Citric acid | • in food preservation • preparing effervescent salts. |
10. The substances which react with acids to form salt and water as only products are called bases. All oxides of metals are bases. All hydroxides of metals are bases.
11. All bases which are soluble in water are called alkalis.
12. Strong bases completely dissociate in aqueous solution. For example, sodium hydroxide (NaOH), potassium hydroxide (KOH), etc.
13. They undergo partial dissociation in aqueous solution. For example, copper hydroxide Cu(OH)2, Zinc hydroxide Zn(OH)2, etc.
14. The bases show the following properties:
(i) These have a bitter taste.
(ii) Bases neither have colour nor small. Only Ammonium hydroxide (NH4OH) has a pungent smell.
(iii) The basic solutions are soapy to touch, i.e., if little amount of sodium hydroxide is rubbed on fingers, it would give a feeling as if soap has been applied on the fingers.
(iv) Bases turn red litmus blue.
(v) Bases contain hydroxide ions e.g., Sodium hydroxide is represented as NaOH, calcium hydroxide is represented as Ca(OH)2, etc., where OH is the hydroxy group.
(vi) Bases react with acids to give salt and water.
15. Uses of Bases
| Bases | Uses |
| (i) Calcium Hydroxide (slaked lime) | • manufacturing of bleaching powder, fungicides, plasters, etc. • neutralising soil acidity. • water softening agent. • antidote for acid poisoning. |
| (ii) Ammonium Hydroxide | • removing grease from • clothes and from window panes. • reagent in laboratory. |
| (iii) Magnesium Hdroxide | • antacid to neutralize stomach acidity. |
16. Special type of substances are used to test whether a substance is acidic or basic. These substances are known as indicators. Turmeric, litmus, china, rose, petals (Gudhal), etc., are some of the naturally occurring indicators.
17. The reaction between an acid and a’base-is known as neutralisation. Salt and water are produced in this process with the evolution of heat.
Acid + Base → Salt + Water (Heat is evolved)
The following reaction is an example :
Hydrochloric acid (HCl) + Sodium hydroxide (NaOH) → Sodium chloride (NaCl) + Water (H2O)
Must Read: