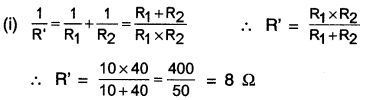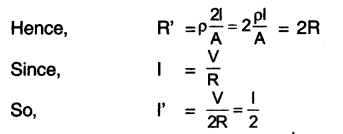HBSE 10th Class Science Important Questions Chapter 9 Heredity and Evolution
Haryana State Board HBSE 10th Class Science Important Questions Chapter 9 Heredity and Evolution Important Questions and Answers.
Haryana Board 10th Class Science Important Questions Chapter 9 Heredity and Evolution
Question 1.
‘Every new generation accumulates variation during reproduction.’ Explain the statement.
Answer:
1. Organisms have two modes of reproduction,
- Asexual reproduction and
- Sexual reproduction. Offspring are born through any of these modes.
2. During reproduction, these offspring inherit two things from their previous generation. They are –
- A common basic body design and
- Some fine (i.e. minor) variations.
3. When the offspring grow and reproduce the next generation, the new generation offspring will again inherit two things.
They are –
- Same basic body design along with differences given by their previous generations and
- Some fine new variations.
4. This way with each new generation born, the variations get accumulated.
![]()
Question 2.
Explain: Parental generation, First generation and Second generation.
Answer:
(a) Parental generation (P):
The first set of parents crossed is called parental generation. The parental generation is denoted by ‘P’.
(b) First generation (F1):
All the offspring born from the first set of parents crossed are said to be belonging to the First generation. The First generation is denoted by F1
(c) Second generation (F2):
All the offspring born from the F1 generation are said to be belonging to the Second generation. The Second generation is denoted by F2.
Question 3.
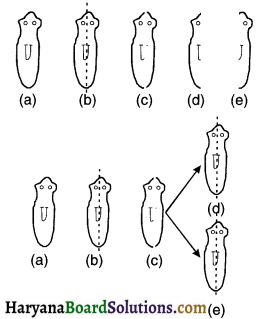
Explain with the help of a diagram how variation (or diversity) is created over succeeding generations.
Answer:
1. As shown in the diagram, consider a single bacteria at the top reproduces on its own asexually.
2. It gives birth to two individuals. The two individuals reproduced will have
- Similar body design like their previous (parental) generation and
- Some minor variations/ differences of their own.
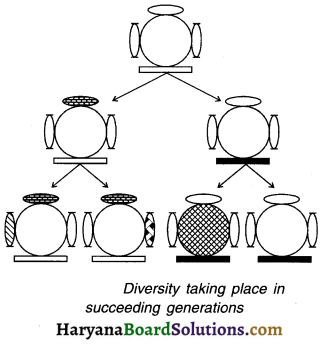
3. Each of these organisms in turn may give birth to two individuals in the next i.e. first generation. So, there are in total four organisms in the second generation.
4. As can be seen, each of the four individuals in the bottom row is somewhat different from each other. Each one has
- The common body design inherited from its previous generation along with
- Some unique differences of its own. So, each organism although similar, is not the exact copy and hence shows variation.
5. Since this is an example of asexual reproduction, the differences are very minor. In case of sexual reproduction, the changes would be much diversed.
![]()
Question 4.
What is the most obvious outcome of reproduction? What do the rules of heredity determine?
Answer:
1. The most obvious outcome of reproductive process is that similar designs of individuals are maintained generation after generation.
2. The rules of heredity determine the process by which traits and characteristics get transferred from one generation to another.
Question 5.
Organisms undergo variation during reproduction. Do all these variations survive?
Answer:
1. Although organisms undergo variation, all the variations do not survive.
2. Depending upon the nature of variations, different individuals would gain different kinds of advantages from the variations.
3. An individual has to withstand several environmental effects and changes. So, organisms having variations which can adapt to the environment survive, others may not.
Question 6.
What do you mean by heredity? Explain.
Answer:
Heredity:
1. Heredity means the transmission of characters from parents to offspring. For example, eggs laid by a sparrow will hatch sparrow and not any other bird. Similarly, a dog gives birth only to pups.
2. Thus, in this sense, it can be said that heredity is continuity of features from one generation to another. This is the essence of heredity.
3. Hereditary information is present in the fertilized egg i.e. zygote.
4. The zygote develops into the same organism to which it belongs. And so the new organisms produced resemble their parents.
![]()
Question 7.
Explain the following terms:
Answer:
1. Heredity: The transmission of parental traits to their offspring is called heredity. This transmission always follows certain principles or laws.
2. Genetics: The study of how inherited traits (or say heredity) are passed-on and the variations occurring therein is called genetics.
3. Trait: A trait is a particular characteristic, quality or tendency that someone or something has. For example, height, skin or eye colour, certain behaviour, etc.
4. Inherited trait: A feature or a characteristic of an organism that it has received in its genes from its parents is called an inherited trait.
Question 8.
Why there are two versions for each trait in each child?
Answer:
In a sexual reproduction, the father as well as the mother contributes equal amounts of genetic material to the child. This means that each trait that the child has can be influenced by the DNA of the father as well as mother. Thus, for each trait there will be two versions in each child.
Question 9.
What are the fundamental characteristics of inheritance of traits?
Answer:
The fundamental characteristics of inheritance of traits are as follows:
(i) A unit of inheritance is called as a gene. Each gene controls a character(s).
(ii) Genes have two alternative forms for each inherited trait.
(iii) From the two alternative traits, the trait which is expressed is known as a dominant trait whereas, the trait which is not expressed is known as a recessive trait.
(iv) From two alternative traits for an expression of a character, one trait may be dominant over the other.
(v) The dominant trait is represented by capital letter (Generally ‘T’) while the recessive trait is represented by the small letter (‘t’).
(vi) The unexpressed/recessive trait may be expressed in any future generation.
(vii) In sexual reproduction both the parents give equal amount of chromatin material/genetic material to make a zygote. So, each inherited trait is influenced by both the parents equally.
![]()
Question 10.
Explain Mendel’s contribution through his study of inheritance of characteristics. OR In Mendel’s study, the ‘T’ gene is dominant while the ‘t’ gene is recessive. Explain in detail with an example. OR Explain Gregor Johann Mendel’s experiment showing inheritance of any one character.
Answer:
1. Mendel studied garden pea plants for the expression of a character. The character under study was the height of plants
2. He observed two contrasting expressions of height — tall plants and dwarf plants/short plants.
3. For the purpose of his study, Mendel took pure tall plants (TT) and pure short plants (tt).
(i) Cross-pollination between parent (P) generation plants:
Mendel performed cross-pollination between TT — pure tall plants and tt — pure short plants. These TT and ft are known as a parent (P) generation plants.
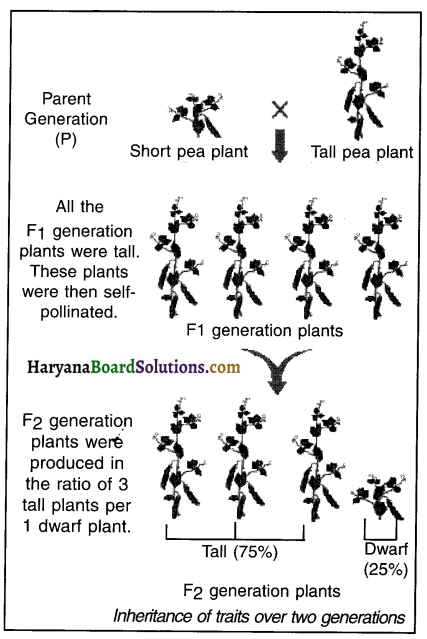
Observation:
The plants that got produced from Parent (P) generation are known as First (F1 generation) plants. Mendel observed that all the plants of F1 generations were as tall as TT of P generation i.e. as tall as their parents. So, Mendel thought that only one parental trait i.e. of tallness (instead of both trait i.e. tallness and shortness) was inherited by the F1 generation plants.
(ii) Self-pollination of (a) Tall Parental Generation plants and (b) F1 generation plants:
In the second phase, Mendel performed self-pollination of
(a) Tall i.e. U Parental Generation plants and
(b) F1 generation plants.
Observation:
(a) The new plants produced by the self-pollination of Tall i.e. U Parental Generation plants were all tall.
(b) The new plants produced i.e. the second (F2) generation plants by the self-pollination of F1 generation plants were a mix of tall and short plants. Mendel observed that in F2 generation, around 75°/° of the plants were tall and 25% of the plants were short. This means the ratio of tall short plant in the F2 generation was 3:1.
Thus, in the second trial, Mendel concluded that in the first trial, the F1 generation plants had inherited both the traits i.e. the trait of tallness and dwarfism, from their parent (P) generation but, only the trait of tallness got expressed. However, when he self-pollinated the F1 generation plants, both the traits i.e. the trait of tallness and dwarfness could be seen in the F2 generation plants.
Question 11.
What did Mendel conclude from his experiment on pea plants?
Answer:
Mendel’s conclusion:
1. The contrasting characters (traits) i.e. tallness and shortness of the pea plant are determined by factors.
2. Two copies (i.e. in pairs) of such factors are present in sexually reproducing organism and they are known as a ‘gene’.
3. These two may be identical, or may be different, depending on the parentage.
4. When individuals having contrasting features are crossed, the feature which gets expressed is known as a dominant feature (trait) and the one that does not get expressed is known as a recessive feature (trait).
5. Tall plants are represented by ‘TT’ as well as ‘Tt’, but short plants are represented by ‘tt’. This means that presence of even single copy of‘T’ is enough to make a plant tall, but for a plant to be short, both the copies have to be ‘t’. Hence, T is called a dominant trait whereas ‘t’ is called a recessive trait.
![]()
Question 12.
Explain Mendel’s experiment on inheritance of two characters simultaneously. OR Explain Mendel’s experiment with different seeds. OR Explain the inheritance of seed colour and shape of the seed of pea plant Pisum Sativum.
Answer:
1. Mendel performed an experiment to study garden pea plants having two different characters. The two characters that the pea plant had were –
- Colour of the seeds and
- Shape of the seeds
2. Mendel wanted to study how the traits express themselves when the plants contain two characters as mentioned above rather than one.
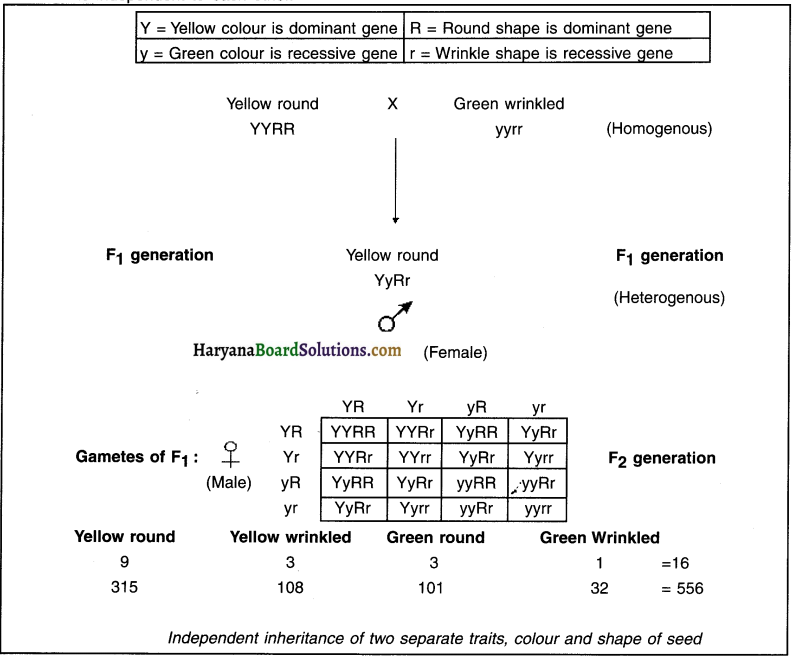
At first, Mendel selected the following two pure breeding pea plants:
(a) Plants with yellow colour seed and round shape seeds.
(b) Plants with green colour seed and wrinkled shape seeds.
- He considered these plants as parent plants.
- Mendel performed cross-pollination between both these parent plants and obtained the F1 generation.
1. Observation of F1 generation plants:
- All the plants of F1 generation had yellow coloured seeds having round shape.
- This means that ‘yellow colour’ and ‘round seeds’ were dominating characteristics (traits) in the F1 generation.
- In the second trial, he performed self-pollination between the F1 plants to obtain F2 generation.
![]()
2. Observation of F2 generation plants:
Mendel obtained four different types of F2 generation plants as mentioned below:
(a) Yellow and round seed plants
(b) Yellow and wrinkled seed plants
(c) Green and round seed plants
(d) Green and wrinkled seed plants
This indicates that characteristics (traits) of colour yellow and green and characteristics (traits) of shape round and wrinkled are inherited independently from each other.
The ratio in which Mendel obtained the plants was —
- (Yellow round) : (Green round) : (Yellow wrinkled) : (Green wrinkled) = 9 : 3 : 3 : 1
- The results obtained in F2 generation clearly indicate that the inheritance of two different characters is independent to each other.
Question 13.
How does the mechanism of heredity work in terms of genes control?
Answer
1. The DNA found in the cells is the fundamental source of genetic information for making specific proteins in the cell.
2. In general, a segment of DNA which provides information to synthesize protein is known as a gene.
3. In plants as well as organisms, the characters (traits) get expressed due to specific reactions. These reactions take place with the help of enzymes.
4. The enzymes as well as several hormones are made up of proteins.
5. So, when a specific gene undertakes synthesis of a specific protein it results in expression of a specific character.
Example:
1. Let us consider height (tallness) as a characteristic of a plant.
2. Plants have hormones that trigger growth. This means height of plants depend upon the amount of a particular harmone.
3. A particular type of enzyme works as a catalyst for this hormone. And the synthesis of this enzyme is regulated by gene located on the DNA.
4. Thus, if the enzyme is synthesized in required amount and if it works efficiently, a lot of quality hormone can be produced which will then boost the height of the plant.
5. However, it the gene has altered due to some reason, the enzyme will become less efficient.
6. This will result in less amount of hormone in the plant and hence it will be of dwarf size.
7. From this example, it can be clearly said that genes control the characteristics or traits.
![]()
Question 14.
Describe sex determination in human beings in detail.
Answer:
Sex determination:
1. In sexual reproduction, the mechanism to determine the sex of an organism is known as sex determination.
2. In human beings, the sex is determined by genes located on the chromosomes.
3. These genes are inherited by the offspring from their parents.
4. In humans, each cell contains 23 pairs of chromosomes.
5. Out of these, 22 pairs are autosomes and one pair is of sex chromosomes.
6. These 22 pairs of autosomes are same in males as well as females. Only the 23 pair differs.
7. In females, the 23 pair contains two similar ‘X’ sex chromosomes i.e. the 23 pair is ‘XX’.
8. In males, the 23rd pair contains one ‘X’ sex chromosome and one ‘Y’ sex chromosome i.e. the 23’’ pair is ‘XV’. The ‘Y’ sex chromosome is smaller than the ‘X’ sex chromosome.
9. Out of all the sperms, 50% sperms contain ‘X’ chromosome while the other 50% sperms contain ‘Y’ chromosome.
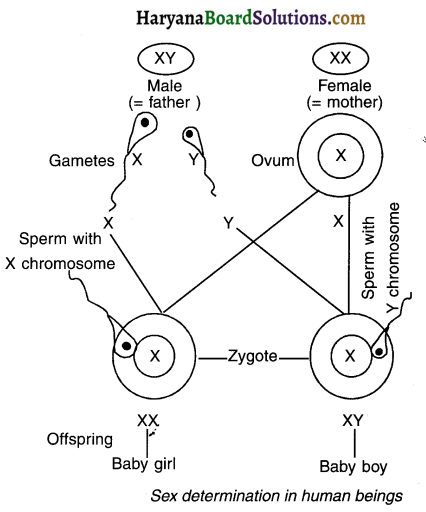
Sex of the foetus:
1. If a sperm carrying ‘X’ chromosome fuses with the ‘X’ chromosome of female egg i.e. if ‘XX’ combination occurs, female will be produced.
2. However, if a sperm carrying ‘Y’ chromosome fuses with the ‘X’ chromosome of female egg, i.e. if ‘XY’ combination occurs, male will be produced.
3. Since male sperm carries both ‘X’ and ‘Y’ chromosomes and female carries only ‘X’ and ‘X’ chromosomes, males are responsible for the sex of the child born.
4. When zygote is formed and the embryo starts developing, the gonads (organs that produce sex cells) are undifferentiated.
5. Later, the gonads develop either into testes or ovaries.
6. If the zygote contains the Y sex chromosomes, the gonad differentiates into testes.
7. These testes then produce male sex hormones and stimulate development of a male individual.
8. Similar process takes place if the zygote contains only ‘X’ chromosome.
Question 15.
What are sex chromosomes? Which sex chromosomes are found in male and female human beings? State the chromosome responsible for the development of male child in human beings.
Answer:
1.The chromosomes that determine the sex of the new born baby are called sex chromosomes. X chromosome and Y chromosome together form the pair of sex chromosomes in male human beings. Thus, the male contains XV sex chromosomes.
2. In female, both the sex chromosomes are X. So, the female sex chromosomes are called XX.
3. A male child can take birth only when the zygote receives Y chromosome from the male human being.
![]()
Question 16.
Every organism resembles its parents. Give reason.
Answer:
1. When male and female sex cells fertilize, zygote is formed.
2. Zygote develops into an organism.
3. Zygote contains heredity information from the parents.
4. This information is passed on to the next generations.
5. Hence, every organism will resemble its parents.
Question 17.
What is natural selection? Explain.
Answer:
1. Natural selection is a central concept of evolution. In general terms it is also called ‘survival of the fittest’.
2. Natural selection can be considered as the one ‘selected by nature’.
3. When organisms develop certain favourable traits i.e. the traits that are more suited for nature to select, the organisms survive and hence reproduce. These organisms then pass their traits to the next generation.
4. Over time this process allows organisms to adapt to their environment. The organisms better adapted to their environment tend to survive and produce more offspring.
Question 18.
Give an example of beetles to understand the concept of natural selection.
Answer:
1. Consider a group of 12 red beetles. They live in bushes mat have green leaves.
2. The population of beetles will grow by sexual reproduction and so every generation produced will show variations.
3. Let us consider that crows feed on these beetles. The more beetles the crows eat, the fewer beetles will remain for reproduction.
4. Now, suppose, a colour variation arises during reproduction. Under this, one of the beetle reproduced attains ‘green colour’ instead of red.
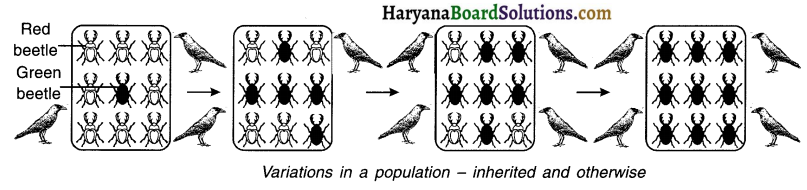
5. When this green coloured beetle undergoes reproduction, it passes this green colour to its progeny. So, all the new beetles reproduced from this green coloured beetle are green.
6. Because green-coloured beetles get merged with green leaves, now the crows cannot see green coloured beetles on the green leaves and therefore cannot eat them. As a result, the green beetles do not get eaten, while the red beetles continue to be eaten.
7. In conclusion, the population of green beetles increase whereas that of red beetles decline drastically.
8. Here, the beetles that adapted the green colour became more suited to nature and so got ‘selected by nature (i.e. natural selection)’ for remaining alive.
![]()
Question 19.
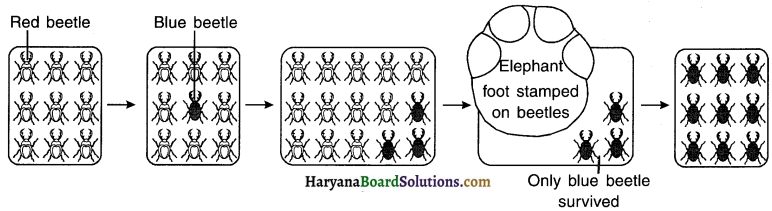
Give an example of beetles to understand the concept of genetic drift.
Answer:
1. Consider a group of 12 red beetles. They live in bushes that have green leaves.
2. The population of beetles will grow by sexual reproduction and so every generation produced will show variations.
3. A colour variation arises during reproduction and few of the new beetles born are of blue colour
instead of red. The blue beetle can pass the colour on to its progeny, so that all its progeny beetles are blue.
4. Crows living in the bush feed on the beetles. They can see both red as well as blue beetles and thereiore can eat both the varieties.
5. Initially, in the population, most of the beetles are of red coloured and only few are blue. At this point, a sudden and an unexpected change takes place. An elephant comes and stamps on the bushes where the beetles live.
6. By chance, the stamping of elephant kills mostly red beetles. The few beetles that have survived are mostly blue.
7. Now, the beetle population again slowly expands, but the beetles in the population are mostly blue.
8. Here, the change in colour as a variation seen in the beetles did not increase their chance of survival, the way it happened when the beetles adopted a green colour. The blue coloured beetle survived simply by chance because the elephant foot got stamped on the red beetles and not the blue.
9. From this we conclude that accidents in small populations can change the frequency of some genes in a population, even if they give no survival advantage This concept is called genetic drift. Genetic drift resulted in variation but did not give any advantage of adaptation to the beetles.
Question 20.
What is genetic drift? Explain.
Answer:
Genetic drift:
The sudden and random change in the gene frequency that occurs by chance in a small population is known as genetic drift.’
Explanation:
1. When a large group of population migrates or dies due to natural calamity, certain traits get eliminated from the population.
2. As a result, in the remaining population the gene frequency is altered. So, in the next progenies some different kind of variations arise.
3. At times due to genetic drift, the population may get free of some unfavourable character and at times, it is also possible that some important character is lost.
Importance:
1. Genetic drift influences evolution.
2. It is possible that the population becomes different because there is a probability that each population may fix a different genotype.
![]()
Question 21.
What is evolution? Explain.
Answer:
Evolution:
1. Evolution is a type of gradual formation of new plants or organisms from the pre-existing primitive plants or animals respectively by Constant and relatively long time changes.
2. The word ‘evolution’ has been derived from Latin word ‘evolvere’ which means to untold’ or ‘unroll’.
3. Evolution is the sequence of gradual changes, which have taken place over millions of years in primitive plants and animals from which new species are formed.
4. Evolution is a constant process taking place in the organisms since life originated. In fact, all the varieties of organisms, which we see around us. have evolved from some ancestors that lived on this earth long time ago.
Question 22.
Explain acquired and Inherited traits (characteristics).
Answer:
(A) Acquired traits:
1. Acquired trait means a trait or characteristic of an organism that it has not inherited but has developed in response to the environment.
2. For example, if an organism starves for sometime and reduces its weight, then it ¡s called acquired trait.
3. The reduced weight due to starvation will not change DNA of the cells.
4. As a result, reduced weight is not a trait, which can be. inherited by future generations of organisms that will starve.
5. Other examples are cut tail of a mouse, a man who knows how to swim, or speak German or roller skate, or may have a scar on the face due to accident. None of these traits will be passed on to the future generations because these traits have not altered the genes of the organisms.
6. Thus, the changes in the non-reproductive body cells of an organism cannot be transmitted to its progeny.
7. Only those traits will be inherited to their progeny where in change has occurred in the genes in gametes of organisms during the process of reproduction.
(B) Inherited trait :
1. A trait of an organism, which is caused by a change in its DNA, is known as inherited trait.
2. For example, suppose a population of red beetles is living in bushes with green leaves.
3. Suppose a colour variation arises during reproduction in the gene of reproductive cells and one green colour beetle arises instead of red.
4. Here, the colour of this beetle is an inherited trait, which can be transmitted to the next generations.
Question 23.
Explain the terms: (a) Variations (b) Acquired variations.
(a) Variations:
- Any difference that occurs between cells, individual organisms, or groups of organisms of any species either by genetic differences or by the effect of environmental factors on the expression of the genetic potential is called variation.
- Organisms may show variation in their physical appearance, metabolism, rate of fertility, mode of reproduction, behaviour, etc.
(b) Acquired variations :
- An acquired characteristic (variation) is a change which is acquired not due to heredity but due to response to the environmental factors.
- Acquired variation may occur in the function or structure of an organism caused after birth due to reasons such as disease, Injury, accident, repeated use or misuse, or other environmental factors.
- Acquired variations are limited to the organism that acquires the variation. Such variations than vanish and with the death of that organism and hence do not get inherited.
![]()
Question 24.
Differentiate between acquired and inherited traits.
Answer:
| Acquired traits | Inherited traits |
| 1. It is a trait of an organism that is not inherited but developed in response to the environment. 2. These changes do not occur in the genes of gametes of the organism. 3. These changes are considered as changes in the non-reproductive body cells. 4. Cut tail of a rat expertise to play a game etc. are acquired characteristics. | 1. It is a trait of an organism, which is caused by a change in its DNA. 2. These changes occur in the genes of the gametes of the organisms. 3. These changes are considered to be changes in the reproductive body cells. 4. Colour of eyes, skin, facial characteristics, etc. are hereditary characteristics. |
Question 25.
What is speciation? Explain.
Answer:
Speciation: The process of evolution of new species that occurs when members of similar population no longer interbreed to produce fertile off spring is known as speciation.
Speciation due to geographical isolation:
1. Suppose due to some reason the population of a given species gets divided into two groups.
2. These groups then get separated from each other geographically by certain barriers like rivers, seas, mountain ranges, or even natural calamities.
3. This means these two groups got geographically isolated and they will not be able to interbreed and hence exchange genes.
4. Over a period, both groups might adapt to its environment through natural selection and develop their gene pool.
5. Eventually, the gene pools of both populations might become so different that the new species of these organisms would evolve in different populations.
6. Thus, due to process of random change in gene frequency (gene drift), after thousands of years individuals become so different that they cannot reproduce with each other and so new species are formed.
7. Thus, geographical isolation leads to reproductive isolation due to which there is no gene flow between two separated groups of population.
8. Thus, we may get completely new species of population.
Other reasons of speciation:
Other reasons can be changes in DNA, change in number of chromosomes, gene cells of two isolated groups of populations, which cannot fuse with each other, etc.
Question 26.
How are organisms classified? OR What do you mean by characteristics as the base of classification? Explain.
Answer:
1. When we classify organisms by grouping them in certain way, it helps us to study them properly.
2. One of the basic methods of classifying the organisms is on the basis of similarities they possess.
3. For classifying we will have to decide which characteristics show ‘fundamental differences’ or say important differences and which show ‘basic differences’ or say less important differences.
Characteristics as a base of classification:
1. A characteristic means a particular type of appearance (form) or behaviour (function).
2. We humans have four limbs (two hands and two legs) and it is our appearance characteristic.
3. Plants perform photosynthesis is a behaviour (function) characteristic.
4. Some basic characteristics are shared by most organisms. For example, cell is the basic unit of life in all organisms. However, the characteristics of the next level of classification would be shared by most, but not all organisms.
5. At cell level, the classification could be whether the cell has a nucleus or not.
For example, cells – of bacteria do not have nucleus, while the cells of most other organisms do.
6. Further, organisms that have nucleus in their cells can be classified as uni-cellular or multi-cellular.
7. Among multi-cellular organisms, classification can be done as whether they undergo photosynthesis or not.
8. The multi-cellular organisms that do not undergo photosynthesis can be further classified as,whether the skeleton is inside the body or around the body.
9. By classifying in this manner we can develop a hierarchy of organisms.
![]()
Question 27.
Giving an example of humans to explain what increases the probability of two species having a common ancestor.
Answer:
The more characteristics two species will have in common, the more closely they will be related. And the more closely they are related, the more are the chances of them being having a recent common ancestor.
Example:
A brother and a sister are closely related. They have common ancestors in the first generation before them, namely, their parents. Now, the girl and her first cousin are also related, but less than the girl and her brother. The reason for this is that although cousins have common ancestors i.e. their grandparents, in the second generation but the parents of these two cousins are different. So, the cousins are less related.
Question 28.
It is believed that non-living material must have given rise to life. How can one say this?
Answer:
1. If we keep on forming small groups of species with recent common ancestors, then eventually we can reach at a stage of forming super-groups of these groups with more distant common ancestors, and so on.
2. Theoretically, if we keep going backwards like this we may reach to a notion that originally there were a single species at the very beginning of evolutionary time.
3. If that is the case, then at some point in the history of the earth, non-living material must have given rise to life.
Question 29.
How did life originate on earth?
Answer:
It is believed that life must have developed from simple inorganic molecules which were present on earth soon after it was formed.
2. It was also speculated that the conditions on earth at that time could have given rise to more complex organic molecules that were necessary for life.
3. It is believed that the first primitive organisms would have arisen from further chemical synthesis.
Question 30.
What do you mean by evidences of evolution? In what way homologous organs and analogous organs give evidence for evolution?
Answer:
Evidence for evolution: Certain significant sources which provide proofs for evolution are called evidences for evolution.
The main ones are:
(1) Evidences through homologous organs,
(2) Evidence through analogous organs and
(3) Evidence through fossils
(1) Evidences through homologous organs:
- Those organs that have same internal structure but different functions are called homologous organs.
- For example, the basic design of internal structure of bones of forelimbs of a frog, lizard, bird, bat and man is same, even though these organs perform different functions.
- This indicates that all these forelimbs have evolved from a common ancestral animal, which had a same basic internal structure.
(2) Analogous organs:
- Those organs, which have different designs but similar appearance and carry out similar functions are called analogous organs.
- For example, wings of insects and birds have different structures but perform similar functions.
- Thus, the presence of analogous organs in different animals provide evidence that they are not evolved from common ancestors, still they perform similar functions to survive prevailing environment.
Question 31.
Differentiate between homologous organs and analogous organs.
Answer:
1. When we classify organisms by grouping them in certain way, it helps us to study them properly.
2. One of the basic methods of classifying the organisms is on the basis of similarities they possess.
3. For classifying we will have to decide which characteristics show fundamental differences’ or say important differences and which show basic differences’ or say less important differences.
Characteristics as a base of classification:
1. A characteristic means a particular type of appearance (form) or behaviour (function).
2. We humans have four limbs (two hands and two legs) and it Is our appearance characteristic.
3. Plants perform photosynthesis is a behaviour (function) characteristic.
4. Some basic characteristics are shared by most organisms. For example, cell is the basic unit of life in all organisms. However, the characteristics of the next level of classification would be shared by most, but not all organisms.
5. At cell level, the classification could be whether the cell has a nucleus or not. For example, cells
of bacteria do not have nucleus, while the cells of most other organisms do.
6. Further, organisms that have nucleus in their cells can be classified as uni-cellular or multi-cellular.
7. Among multi-cellular organisms, classification can be done as whether they undergo photosynthesis or not.
8. The multi-cellular organisms that do not undergo photosynthesis can be further classified as, whether the skeleton Is Inside the body or around the body.
9. By classifying in this manner we can develop a hierarchy of organisms.
![]()
Question 27.
Giving an example of humans to explain what increases the probability of two species having a common ancestor.
Answer:
The more characteristics two species will have in common, the more closely they will be related. And the more closely they are related, the more are the chances of them being having a recent common ancestor.
Example:
1. A brother and a sister are closely related. They have common ancestors in the first generation before them, namely, their parents.
2. Now, the girl and her first cousin are also related, but less than the girl and her brother. The reason for this is that although cousins have common ancestors i.e. their grandparents, in the second generation but the parents of these two cousins are different. So, the cousins are less related.
Question 28.
It is believed that non-living material must have given rise to life. How can one say this?
Answer:
1. It we keep on forming small groups of species with recent common ancestors, then eventually we can reach at a stage of forming super-groups of these groups with more distant common ancestors, and so on.
2. Theoretically, if we keep going backwards like this we may reach to a notion that originally there were a single species at the very beginning of evolutionary time.
3. It that is the case, then at some point in the history of the earth, non-living material must have given rise to life.
Question 29.
How did life originate on earth?
Answer:
1. It is believed that life must have developed from simple inorganic molecules which were present on earth soon after it was formed.
2. It was also speculated that the conditions on earth at that time could have given rise to more
complex organic molecules that were necessary for life.
3. It is believed that the first primitive organisms would have arisen from further chemical synthesis.
Question 30.
What do you mean by evidences of evolution? in what way homologous organs and analogous organs give evidence for evolution?
Answer:
Evidence for evolution: Certain significant sources which provide proofs for evolution are called evidences for evolution.
The main ones are:
(1) Evidences through homologous organs
(2) Evidence through analogous organs and
(3) Evidence through fossils
(1) Evidence.s through homologous organs:
- Those organs that have saine internal structure but iterent functions are called homologous organs.
- For example, the basic design of internal structure of bones of forel,mbs of a frog, lizard, bird, bat and man is same. even though these organs perform different functions.
- This Nidicates that all these forelimbs have evolved from a common ancestral animal, which had a same basic internal structure.
(2) Analogous organs:
- Those organs, which have different designs but similar appearance and carry out similar functions are called analogous organs
- For example, wings of insects and birds have different structures but perform similar functions
- Thus, the presence of analogous organs in different animals provide evidence that they are not evolved from common ancestors still they perform similar functions to survive prevailing environment.
![]()
Question 31.
Differentiate between homologous organs and analogous organs.
Answer:
| Homologous organs | Analogous organs |
| 1. These organs have similar internal structure. 2. These organs perform different functions. 3. These organs have evolved from a common ancestor. 4. For example, forelimbs of a frog, lizard, a bird, bat and a man are homologous | 1. These organs have different basic design. 2. These organs perform similar functions. 3. These organs have not evolved from a common ancestor. 4. Wings of insects and birds are examples of analogous organs. |
Question 32.
What are fossils? How do they provide evidence for evolution? OR What are fossils?
Answer:
1. The remains of dead organisms buried under the earth for millions of years are known as fossils.
2. Fossils are impressions of dead plants or animals that lived in the past.
3. When plants or animals die, the micro-organisms get decomposed in the presence of moisture and oxygen.
4. However, sometimes due to environmental conditions, their bodies do not decompose completely.
5. Such body parts of the plants and animals become fossil and can be available on digging the earth.
6. Fossils can be in the form of imprints, burrow of a worm, or even an actual bone.
Example:
- If the dead leaf gets caught in the mud, leaf will not decompose completely.
- The mud around the leaf will set around it as a mould which will then slowly become hard to form a rock and retain the impression of the leaf. Thus fossil of a leaf is formed.
- By studying these fossils, scientists learn how organisms evolved over time.
Question 33.
Describe any three methods of tracing evolutionary relationships among organisms.
Answer:
Evidence for evolution: Certain significant sources which provide proofs for evolution are called evidences for evolution.
The main ones are:
(1) Evidences through homologous organs
(2) Evidence through analogous organs and
(3) Evidence through fossils
(1) Evidences through homologous organs:
- Those organs that have saine internal structure but different functions are called homologous organs.
- For example, the basic design of internal structure of bones of forel,mbs of a frog, lizard, bird, bat and man is same. even though these organs perform different functions.
- This Nidicates that all these forelimbs have evolved from a common ancestral animal, which had a same basic internal structure.
(2) Analogous organs:
- Those organs, which have different designs but similar appearance and carry out similar functions are called analogous organs
- For example, wings of insects and birds have different structures but perform similar functions
- Thus, the presence of analogous organs in different animals provide evidence that they are not evolved from common ancestors still they perform similar functions to survive prevailing environment.
1. The remains of dead organisms buried under the earth for millions of years are known as fossils.
2. Fossils are impressions of dead plants or animals that lived in the past.
3. When plants or animals die, the micro-organisms get decomposed in the presence of moisture and oxygen.
4. However, sometimes due to environmental conditions, their bodies do not decompose completely.
5. Such body parts of the plants and animals become fossil and can be available on digging the earth.
6. Fossils can be in the form of imprints, burrow of a worm, or even an actual bone.
Example:
- If the dead leaf gets caught in the mud, leaf will not decompose completely.
- The mud around the leaf will set around it as a mould which will then slowly become hard to form a rock and retain the impression of the leaf. Thus fossil of a leaf is formed.
- By studying these fossils, scientists learn how organisms evolved over time.
![]()
Question 34.
Mention three important features of fossils which help in the study of evolution.
Answer:
(i) Fossils represent how the ancient species were preserved.
(ii) Fossils help in establishing evolutionary relationship between organisms and their ancestors.
(iii) Fossils help in knowing the time period in which organisms lived.
Question 35.
How can you say that eyes evolved in stages?
Answer:
1. The eye is an important organ for animals.
2. It is a complicated organ which cannot be generated by a single DNA change.
3. The eyes of animals have been created in stages after many generations.
4. First of all, eye was formed in planaria (flat-worm).
5. The eyes of planaria are very simple and are just like ‘eyes-spots’ which detect light.
6. These simple eyes provide a survival advantage to planaria.
7. Thus eye seems to be a very popular adaptation. Gradually, it becomes a complex organ.
8. Most of the animals like insects, octopus, invertebrates and vertebrates have eyes.
9. The structure of eye in above organisms is different which suggests the evolution of eye and is an example of evolution taken place stages.
Question 36.
How can you say that birds are very close to reptiles? OR Give reason. Birds are closely related to reptiles.
Answer:
1. In some dinosaurs, feathers could not be used for flying but provided insulation in cold weather. But, later they might have become useful for flight. Birds however later adapted to flights.
2. Thus, presence of feathers in the birds tells us that birds are quite closely related to reptiles, since dinosaurs which had feathers were reptiles.
Question 37.
Explain how wild cabbage has evolved over the years. OR Explain evolution by stages of analogous organs with the help of artificial section of evolution method.
Answer:
1. As per the definition of analogous organs, the organs that have different basic design but similar appearance and carry out similar functions are called analogous organs.
2. From this concept, it is said that very dissimilar looking structures evolved from common ancestral body design. But all these guesses are about history, which is very old.
3. A present day example to prove this concept can be of wild cabbage.
4. If we study cabbage, we can learn that cabbage provides evidence that a completely dissimilar ‘ looking plant can evolve from it by the process of evolution.
5. Before more than 2000 years, the farmers cultivated wild cabbage as food plant.
6. With the passage of time, farmers gave various modifications to it.
7. Some farmers obtained broccoli (a kind of vegetable) while others obtained sterile flowers and developed cauliflower.
8. Some other farmers selected swollen parts of wild cabbage and developed another variety known as kohlrabi.
9. Some farmers have developed slightly large leaves of wild cabbage and their leafy vegetable is called kale.
10. Thus, now wild cabbage is the ancestor of cabbage, broccoli, cauliflower, kohlrabi and kale varieties, which are obtained by farmers by performing artificial selection. Moreover, all these varieties look different from their ancestor i.e. wild cabbage.

Question 38.
It is incorrect to consider evolution as progress. Explain.
Answer:
1. Actually, no real ‘progress’ has taken place in the idea of evolution.
2. Evolution is simply the generation of diversity and the shaping of the diversity by environmental selection.
3. The only progressive trend that is seen in evolution is that with time more and more complex body designs have emerged. This does not mean that the older designs were inefficient. Many older and simpler designs still survive.
4. One of the simplest life forms bacteria still inhabit the most inhospitable habitats like hot springs, deep-sea thermal vents and the ice in Antarctica.
5. Hence, it is incorrect to consider evolution as progress.
![]()
Question 39.
How can you say that humans have not evolved from different races and that they all belonged to only one region of the world?
Answer:
1. Across the earth, there is a great diversity in humans.
2. For quite a long time, man thought about different races of human.
3. Initially, human race was identified on the basis of skin colour and the humans were known as yellow, black, white or brown.
4. However, in the recent years, it has been proved that all the human beings have evolved from a single species called the Homo sapiens.
5. By research, it has been established that we all have emerged from Africa.
6. Our genetic foot prints can be traced back to Africa.
7. A couple of hundred thousand years ago, some of our ancestors left Africa while others stayed back.
8. Those who left Africa slowly spread over the planet from Africa to West Asia to Central Asia, Eurasia, South Asia and East Asia.
9. Humans also migrated to islandsof Indonesia and Philippines, Australia and reached America.
10. They went with groups, sometimes separating from each other and mixing with each other, even moving in and out of Africa.
11. Thus, humans have all evolved from Homo sapiens and initially belonged to Africa.
Question 40.
Differentiate between dominant trait and recessive trait.
Answer:
| Dominant trait | Recessive trait |
| These traits can be seen in F1 generation. Dominant trait is found in F1 as well as F2 generation in larger quantities. F1 generation is a cross of two plants with different traits. | These traits cannot be seen in F1 generation. Recessive trait is found in F2 generation, that too in lesser quantity. F2 generation is a self-pollination of F1 generation. |
Question 41.
Give the pair of contrasting traits of the following characters in pea plant and mention which is dominant and which recessive.
(i) Yellow seed
(ii) Round seed
Answer:
| Character | Contrasting traits | |
| Dominant | Recessive | |
| (i) Seed colour (ii) Seed type | yellow round | green wrinkled |
Question 42.
Why did Mendel choose pea plant for his experiments? OR Mendel selected pea plant (Pisum sativum) for his experiments to study inheritance. Give reason.
Answer:
1. Pea plants are very small and so it is quite easy to manage them. Also, they grow easily.
2. They produce a large number of progeny and so the characters they show can be studied more accurately on a larger scale.
3. Pea plants have both male and female reproductive organs. As a result, they can either self-pollinate themselves or cross-pollinate with another plant. This helps in understanding the traits properly.
4. Pea plants have sharply defined and contrasting traits such as height (tall, dwarf), seed colour (yellow, green), etc.
5. They are annual plants. So, every year, one can obtain a new generation plant.
6. These characteristics of the pea plant make it quite favourable for performing research. Hence, Mendel choose this plant.
![]()
Question 43.
Males are responsible for the sex of the child produced. Give reason.
Answer:
1. Sex of the child is determined by the sex chromosomes that its zygote contains.
2. If the zygote contains ‘Y’ sex chromosome over and above ‘X’ chromosome, it will turn into male and if both sex chromosomes are ‘X’, the child born will be a female.
3. A female sex cell contains ‘XX’ sex chromosome whereas a male contains ‘XY’ chromosome.
4. Thus, a female can always donate ‘X’ chromosome but it is the male that can provide ‘Y’ chromosome to female ‘X’ chromosomes and produce a male.
5. However, if the male donates ‘X’ chromosome, a female will be born.
6. Thus, males and not females are responsible for the sex of the child produced.
Question 44.
Changes in the non-reproductive body cells of an organism cannot be transmitted to its progeny. Give reason.
Answer:
1. Only those traits will be inherited to their progeny wherein change has occurred in the genes in gametes of organisms during the process of reproduction.
2. For example, if an organism starves for some time and reduces its weight, then It is called acquired trait.
3. The reduced weight due to starvation will not change DNA of the cells.
4. As a result, reduced weight is not a trait, which can be inherited by future generations of organisms that will starve.
5. Thus, the changes in the non-reproductive body cells of an organism cannot be transmitted to its progeny
Question 45.
Does genetic combination of mothers play a significant role in determining the sex of a new born?
Answer:
1. The mothers have a pair of XX chromosomes. This means that all children whether boy or girl can inherit X-chromosome only from their mother.
2. Only males have XV chromosome. One of these two will be passed on to the offspring. So, whether the sex of the new born will be male or female can be decided only through the male chromosome. Hence, genetic combination of mothers does not play a significant role in determining sex of the new born.
Question 46.
Why do all the gametes formed in human females have only X-chromosome?
Answer:
The genetic making of the females is such that their sex chromosomes have only X chromosomes. This means both the chromosomes in the female germ cells have X chromosome. So, naturally, the gametes formed in human females have only X-chromosomes.
Question 47.
A woman has only daughters. Analyze the situation genetically and provide a suitable explanation.
Answer:
1. Man contains both X and Y chromosomes. This means males have XV chromosome. So, it is mans sex cells that determine the sex of the child born.
2. Women contain only XX chromosomes so she can contribute X chromosome only. A woman has only daughters. This indicates that in every fusion, the sperm of the male carrying X-chromosome got fertilized with the X chromosome of the female ovum. So, if a boy were to be born, the Y chromosome from male should have travelled and fertilized with X chromosome of the female.
![]()
Question 48.
Why reduction in weight of beetle bug cannot be considered as an inherited trait?
Answer:
1. The weight of the beetle bug got reduced because of starvation. It did not change the DNA of the germ (sex) cells. Change in non-reproductive tissues cannot be passed on to the DNA of the germ cells.
2. Since the DNA is not changed, the change i.e. low weight cannot be inherited by the progeny of a starving beetle.
3. Hence, even if some generations of beetles are low in weight because of starvation, it cannot be considered as an example of evolution because the change is not inherited over generations. When such scarcity of food gets over and the beetle get sufficient food the new beetles will be healthy.
Question 49.
‘Experience of an Individual during its lifetime cannot be passed on to its progeny and cannot direct evolution’ What do you mean by this statement? Explain giving example.
Answer:
Change in non-reproductive tissues cannot be passed on to the DNA of the germ cells. Hence the experiences of an individual during its lifetime can neither be passed on to its progeny nor can such experiences result in evolution.
Example:
1. If we breed a group of mice, all their progeny will have tails.
2. Suppose we remove the tails of each generation of these mice by surgery then it does not mean that the progeny is tailless.
3. Artificially removing the tail does not bring any change in the genes of the germ cells of the mice.
4. So, the cut-tall that a generation of mice experienced cannot become a trait to be passed on to the next generation.
Question 50.
Work out which trait would be considered dominant and which one recessive in the given figure.
Answer:
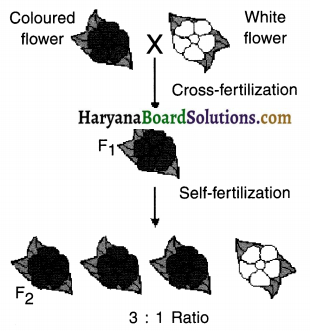
Stage 1 (F1 generation):
When a cross occurs between a coloured flower plant and a white flower plant, the F1 generation plant produced has all the coloured flowers. This means ‘the coloured flowered trait’ is dominant over ‘white flowered trait’.
Stage 2 (F2 generation):
Although, the white flowered trait did not get expressed in F1 generation. it did get expressed in F2 generation. This means the ‘white flowered trait’ is the recessive trait.
Question 51.
Sakhaaram, a poor farmer has 2 daughters. He feels that he should have a son who can help him in his farming work. So when his wife again gets pregnant, he tells her that this time she should not give birth to a girl again. If she does so, he will abandon her. So, she insists her to go for sonography and remove foetus it is found to be of a female.
Now, answer the following questions:
![]()
Questions:
1. Who is the responsible for the sex of the children? — Sakhaaram or his wife?
2. ‘Owing to the poor condition of Sakhaaram, removing the female foetus is a good solution.’ State your comments.
3. Which sex chromosome of Sakhaaram did not do its job of giving birth to a male child in previous cases?
4. The village sarpanch advised Sakhaaram to accept the born child irrespective of the sex as it is God’s decision and also because that is the only legal way. Which values has Sarpanch shown?
Answers:
1. Sex of the child gets determined by the chromosome of the male parent. Hence, Sakhaaram is responsible for the sex of the children.
2. Removing female foetus is a crime. Sakhaaram can be jailed for doing so. Moreover, socially, such an act disturbs the sex-ratio.
3. Y-chromosome.
4. The Sarpanch has shown the values of morality, empathy towards Sakhaaram and care and concern towards his wife. Moreover, the Sarpanch also displayed the values of awareness towards the laws of our country.
Question 52.
Given below is the experiment carried out by Mendel to study inheritance of two traits in garden pea:
(a) What do the letters A, B and C represent?
(b) State the objective for which Mendel performed this experiment.
Answer:
(a) A = gamete of round green (RY) plant
B = gamete of wrinkled (ry)
C = F1 generation (all round yellow)
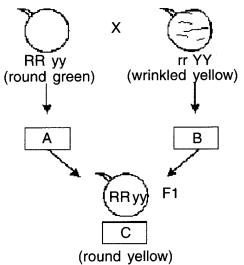
(b) Mendel’s objective was to show independent inheritance of traits or to prove law of independent assortment.
Question 53.
The legendary Milkha Singh, also known as ‘The Flying Sikh’ is the source of inspiration for millions of indians who aspire to become a great runner. However, Milkha’s son Jeev is a golfer and not a runner.
Now answer the following questions:
Questions:
1. Didn’t Jeev inherit the running skill from Milkha Singh? Why or why not?
2. Which type of traits is running?
Answers:
1. Skills such as running, swimming, acting, dancing, etc. cannot be inherited because such skills are acquired skills. Milkha Singh acquired it by extreme hard work and dedication. His son acquired the skills of golfing.
2. Running is an acquired skill. It can be acquired by putting dedicated and planned effort in the field of one’s choice.
Question 54.
Explain whether the traits like eye colour or height are genically Inherited or not. Do power to lift weight and reading French also belong to same category? Justify your answer.
Answer:
(a) Colour of eye and height are genetically inherited.
Reason:
Traits such as colour of eye and height cannot be changed or acquired during a life because they are controlled by the genes which an individual inherits from the parents.
(b) Power to lift weight and reading French can be acquired during life time.
Reason:
1. These traits are achieved by an individual by experience and practice. Hence, these are acquired characters.
2. These acquired characters do not change DNA of germ cells and so they cannot be inherited or passed to the next generation.
![]()
Question 55.
Which of the following traits cannot be passed on the progeny? Justify your answer.
(i) Rudimentary eyes of planaria.
(ii) Absence of tail in a mouse (after surgical removal)
(iii) Low weight of a starving beetle.
Answer:
(i) Trait number
(ii) Absence of tail in a mouse (after surgical removal) and
(iii) Low weight of a starving beetle cannot be passed on the progeny.
Reason:
These traits are present in non-reproductive tissues. So, they do not change the DNA of the germ cells.
Very Short Answer Type Questions
Question 1.
Define heredity.
Answer:
The process of transmission of characters from parents to the offspring is known as heredity.
Question 2.
Define genetics
Answer:
The branch of science which deals with heredity and variation is known as genetics
Question 3.
In a sexual reproduction, offspring is similar to the characteristics of parents however it is not identical to the parents. Justify this statement.
Answer:
Inheritance from previous generation includes carrying forward the basic body structure along with minor variations. Hence It is rightly said that though offspring is similar it is not identical to the parent.
Question 4.
Why would you say that asexual reproduction is not suitable for introducing variation in species?
Answer:
Asexual reproduction involves a single parent. The offspring produced has same set of chromosomes which exhibit little or no variation. Hence asexual reproduction is not suitable for introducing variation in species.
Question 5.
How can we say that variation In species Increases the probability of survival?
Answer:
Variation improves the adaptability of the species according to the climatic conditions. This increases the chances of survival.
Question 6.
State the types of earlobes that can be seen in humans.
Answer:
We can see two types of earlobes in humans. They are —
(i) Attached earlobes Le, earlobes which are attached to the side of the head.
(ii) Free earlobes i.e. earlobes which seem hanging and hence unattached to the side of the head.
Question 7.
Point out a variation that has been observed in human beings.
Answer:
Free or attached earlobes is a variation that can be observed in human beings.
![]()
Question 8.
What is meant by a pure plant (or pure line plant)?
Answer:
A breed or strain of animals or plants that maintains a high degree of consistency in certain characters as a result of inbreeding for generations is called a pure breed of plant or animal. Such a plant of animal maintains homozygous for all genes.
Question 9.
Who is considered as a ‘father of genetics’?
Answer:
Gregor Johann Mendel is considered as a ‘father of Genetics’.
Question 10.
What is the contribution of Gregor Johann Mendel?
Answer:
Gregor Johann Mendel established Mendelian Inheritance Laws. He made major contributions in genetics.
Question 11.
Which term did Mendel use to describe genes?
Answer:
Mendel used the word ‘factor’ to describe genes.
Question 12.
Name the plant on which Mendel performed his experiments.
Answer:
Mendel performed experiments on Pisum Sativum (Garden pea)
Question 13.
What do you mean by dominant and recessive traits?
Answer:
While inheriting the traits from parent, the allele of a gene that masks the other variants is called as the dominant trait whereas the one that gets masked is called as recessive trait.
![]()
Question 14.
What are F1 and F2 generations?
Answer:
F1 is the first generation reproduced from two distinct parental type of same species. F2 is the second generation reproduced from breeding of F1 generation.
Question 15.
What did Mendel observe in F1 progeny when he crossbred a tall and a short plant?
Answer:
Mendel observed that there were no medium height plants. All the plants in F1 progeny were tall in height.
Question 16.
What was the second observation that Mendel make from F2 progeny of garden peas plant?
Answer:
Mendel found that 75% of F2 generation plant produced from F1 progeny were tall whereas 25% of them were short heighted.
Question 17.
If TT and Tt trait combination produces tall plants, which of the traits is a dominant trait?
Answer:
In the above stated combination of traits, T is the dominant trait for tallness.
Question 18.
How did Mendel prove that the traits are independently Inherited by the progeny?
Answer:
When Mendel cross pollinated one plant with yellow and round seeds with another plant having green and wrinkled seeds, he observed that the F2 progeny generated four different types of plants with the different combinations of the above mentioned traits. This proves that the traits are independently inherited by progeny.
Question 19.
What is the phenotype ratio of Mendel’s experiment done to study inheritance of a single character?
Answer:
3:1
Question 21.
What is the genotype ratio of Mendel’s experiment done to study InherItance of a single character?
Answer:
1:2:1
Question 22.
What is the phenotype ratio of Mendel’s experiment to study inheritance of two characters?
Answer:
9:3:3:1
![]()
Question 23.
State the correct sequence for the expression of tall trait in pea plant.
(A) A lot of hormone will be made
(B) A gene provides information for protein
(C) A hormone triggers the growth in height
(D) An enzyme works efficiently
Answer:
(B) – (D) – (A) – (C)
Question 24.
Write expanded form of DNA.
Answer:
Deoxyribonucleic acid
Question 25.
What is a gene?
Answer:
Gene is considered to be the basic unit of heredity. A section of DNA in a cell which provides information of a protein is called as gene for that protein.
Question 26.
How is the sex of an offspring determined in human beings?
Answer:
The genes inherited from the parents determines the sex of the offspring in human beings. If an X chromosome is inherited from father, the sex will be female whereas if Y chromosome is inherited from father, the sex will be male.
Question 27.
Give an example of an animal in which sex is not genetically determined.
Answer:
In snails, sex is not determined genetically.
Question 28.
Would a new born child with XX pair of chromosome be a boy or a girl?
Answer:
The new born child with XX pair of chromosome would be a girl.
Question 29.
What is the difference between the sex chromosome of a male and a female?
Answer:
Women have a perfect pair of sex chromosomes and both are called X whereas men have a mismatched pair in which one is X with a normal length and the other is Y which is of shorter length.
![]()
Question 30.
Suppose, a gene of brown eyes is dominant to a gene of blue eyes. What would be the eye colour of a child born If he inherits gene of blue eyes from mother and a gene of brown eyes from father.
Answer:
The child have brown coloured eyes.
Question 31.
It a woman with brown hair gene b marries a man with black hair gene B, what would be the hair colour of offspring born with the below mentioned combination?
(i) bb, (ii) Bb, (iii) BB
Answer:
Offspring born with contrition bb would have brown hair whereas the offspring born with the combinations Bb and BB would have black hair
Question 32.
How would you differentiate an acquired trait from an inherited trait?
Answer:
The trait of an organism that is passed on from the DNA of a parent to an offspring is an inherited flit whereas the trait that an organism develops as a response to the environment and through me experience is an acquired trait.
Question 33.
Do young once of mice whose tails have been cut are born without tails? Justify your name.
Answer:
The young ones are born with tails because cutting the tail of parent mice does not after the genes of mice
Question 34.
What is evolution?
Answer:
The change introduced in the offspring from the parents through variation in species is known as evolution.
![]()
Question 33.
Name the scientist who first came up with the theory of evolution.
Answer:
Changes Darwin first introduced the theory of evolution.
Question 38.
Which are the two basic factors responsible for evolution?
Answer:
Heredity and deflation are the two basic factors responsible for evolution.
Question 37.
What are the two population of one species who cannot reproduce called as?
Answer:
Such poputation are called as independent population.
Question 38.
What is natural selection?
Answer:
The process whereby organisms get batter adapted to their environment tend to survive and produce more oftsprng is called …….. .
Question 39.
State any three reason which cause speciation.
Answer:
Geographical isolation, changes in DNA. emergence of a variation n population. etc are few of the newly reasons which can cause speciation.
Question 40.
How can geographical isotation impact specistion in species which reproduce:
(i) asexualy,
(ii) sexually?
Answer:
In case of asexual reproduction geographical isolation may not result in speciation because the offspring are reproduced from already existing parent throught salt fertilization. it has high chance of speciation in case of sexual reproduction as geoephically isolated species may rarely get involved in cross fertilization.
![]()
Question 41.
What do you mean by genetic drift?
Answer:
The sudden and random changes in the gene frequency occurring in a small population by chance is known as a genetic drift.
Question 42.
I am able to inculcate diversity in organisms without any adaptations that too simply by chancing the frequency of certain genes in a population. identify me.
Answer:
Genetic drift
Question 43.
On what basis do we classify organisms? Give an example.
Answer:
We can classify organisms based on characteristics i.e. the appearance or behavior. The ability to do photosynthesis is a characteristic which helps us classify these organisms in plant kingdom.
Question 44.
What are the homologous characteristics of organisms? Give an example.
Answer:
The organs having similar origin and similar basic structure but may perform different functions are homologous organs. For example, frogs. lizards, birds and humans. All of these have limbs with similar structures but they are used to perform different functions.
Question 45.
Describe analogous organs with an example.
Answer:
Analogous organs are the organs which have similar functions even though they might have different origin. For example, the wings of a bat and a butterfly perform the functions of flying but they do not have common ancestry.
![]()
Question 46.
What are fossils?
Answer:
When the organisms die, their body gets decomposed and then gets absorbed in nature. But at times, the body or few parts of body remains without getting decomposed completely. Such preserved traces of organisms are called fossils.
Question 47.
Which evolutionary explanation is true for the feathers?
(1) Birds are very closely related to reptiles
(2) Birds and bat are very closely related to each other.
Answer:
Statement I
Question 48.
What Is the ‘relative way’ to find the age of a fossil?
Answer:
Relative way compares the depth of two fossils at which they are found in earth’s surface. The fossil that is found near to surface of earth is more recent than fossil that is discovered at deeper layer.
Question 49.
Which fossil dId we discover in Narmada Valley recently?
Answer:
A skull of a dinosaur was discovered in Narmada valley a few years ago.
Question 50.
How does the dating method work for calculating the age of a fossil?
Answer:
In dating method, the different isotopes (Carbon-14) of the same element found in fossil are compared to the current available element for calculating the age of a fossil.
Question 51.
What is the possible explanation of evolution of complex organs like eyes?
Answer:
It can be explained that the evolution of such complex organs took place at regular stages. For example, it may have started with a rudimentary eye which initially just detected light.
Question 52.
Give an example of an animal having rudimentary eye.
Answer:
Planaria is a type of flat-worm having rudimentary eye.
Question 53.
Mention one characteristIc which lndicates close relation between birds and reptiles
Answer;
Existence of feathers on the body of few dinosaurs (who were reptiles) and also on the body of birds indicates a close relationship between birds and reptiles.
Question 54.
Give an example of evolution through artificial selection. OR List any six vegetables which have been evolved from wild cabbage due to artificial selection.
Answer:
Humans, through artificial selection have cultivated wild cabbage by focusing on various characteristics. This resulted in evolution to multiple types of vegetables such as cauliflower, broccoli, kohlrabi, kale, cabbage and red cabbage.
Question 55.
How can variation in population help us in understanding evolution?
Answer:
We know that minor changes in DNA during reproduction is the basic event In evolution. If we compare DNA of different species over the period of time this can help us in understanding evolution of species.
Question 56.
Give an example which proves that older body designs and characteristics are not in efficient.
Answer:
Bacteria, which are the simplest and oldest body designs are successfully surviving in conditions such as hot springs, deep-sea thermal vents and ice in Antarctica. This proves that older body designs and characteristics are not inefficient.
![]()
Question 57.
Where were the earliest traces of human species found? What is the name of that species?
Answer:
The earliest traces of human beings known as Homo sapiens have been found in Africa.
Fill in the Blanks:
1. During reproduction, an offspring inherits two things from its previous generation, common basic body design and …………..
Answer: some fine (minor) variations
2. Variations in the new generation occur due to ……………
Answer: DNA copying
3. Selection of variants by environmental factors forms the basis for …………………
Answer: evolutionary processes
4. The rules of determine the process by which traits and charactenstics are reliably inherited.
Answer: Heredity
5. If the traits get expressed in the first generation, they are known as …………..
Answer: Dominant traits
6. The DNA segment which is responsible for the synthesis of a specific type of protein is called ……………..
Answer: Gene
7. The lowest part of the ear pinna is known as an …………..
Answer: Ear-lobe
![]()
8. …………… can change their sex on their own.
Answer: Snails
9. It is the chromosome of ……………….. that determines the sex of the child born.
Answer: male sex cell
10. In layman language, the phrase related to natural selection is ……………..
Answer: survival of the fittest
11. A trait developed in response to the environment is called ………………..
Answer: acquired trait
12. Acquired traits are ……. in nature.
Answer: temporary
13. Genetic drift leads to accumulation of …………………
Answer: different changes (variation)
14. The of eyes provide survival advantage to them……………..
Answer: planaria
15. Thus, presence of feathers in the birds tells us that birds are quite closely related to…………
Answer: reptiles
16. …………. used to be the most common way of identifying human races.
Answer: Skin colour
True OR False
1. Organisms have two modes of reproduction. — True
2. Suppose a bactena is able to survive better in a heat wave. This means that quite likely the next generation will be able to survive in a heat-wave — True
3. Organisms undergo variation and most variations survive resulting in high rate of survival of those organisms. — False
4. If a trait A exists in 8% of a population of an asexually reproducing species and a trait B exists in 42% of the same population, it means population with trait A must have arisen earlier and is more stable to variation. — False
5. Practically, the father contributes more amount of genetic material to the child as compared to the mother. — False
6. In Mendel’s experiment, in the first place, no ‘medium-height’ plants were produced. This means the plants gained trait only from one of the parent and not both and so his first experiment failed. — False
7. In Mendel’s experiments F1 progeny was obtained by cross pollination. — True
8. In Mendel’s experiments F2 progeny was obtained by cross pollination. — False
9. In Mendel’s experiments, characters of both the parents were observed in F2 generation. — True
10. The enzyme synthesis in the cell is controlled by genes. — True
11. In general, the pea plant shows cross-pollination. — True
12. In some reptiles, change in temperature of the fertilized egg determines the sex of the newborn. — True
13. The size of both the chromosome in women is same, while it is unequal in men. — True
14. A child who inherits a Y chromosome from her father will be a girl, and one who inherits an X chromosome from him will be a boy. — False
15. If a person is on dieting and loses weight, he is said to have acquired a trait. — True
16. An inherited trait alters the DNA. — True
17. If a person from Ahmedabad who loves living in mountains migrates to Himalayas and lives there for 20 years, his child when born will develop similar love for the mountains. — False
18. Our forelimbs are similar to lizards. — True
19. It is believed that both, human beings and chimpanzees have a common ancestor. — True
20. Practically speaking, there is no real ‘progress’ in the idea of evolution. — True
Match the Following:
Question 1.
| Column I. | Column II. |
| 1. Gene 2. New combination 3. Y-chromosome 4. Inheritance | p. Genetical trait q. Segment of DNA r. Mitosis s. Male sex |
Answer:
(1- q), (2 – r), (3 – s), (4 – p)
Question 2.
| Column I. | Column II. |
| 1. Ammonites 2. Snail 3 Planaria 4. Dinosaurs having feathers | p. Reptilia q. Eye-spots for the first time r. Can change sex s. Fossils |
Answer:
(1 – s), (2 – r), (3 – q), (4 – p)
![]()
Question 3.
| Column I. | Column II. |
| 1. Variation 2. Natural selection 3. Artificial selection 4. Dating fossils | p. Ratios of isotopes q. Change in form or function of the organism r. Change in variety through selective hybridization s Evolution of species |
Answer:
(1 – q), (2 – s), (3 – r), (4 – p)
HBSE 10th Class Science Important Questions Chapter 9 Heredity and Evolution Read More »