Haryana State Board HBSE 10th Class Science Important Questions Chapter 2 Acids, Bases and Salts Important Questions and Answers.
Haryana Board 10th Class Science Important Questions Chapter 2 Acids, Bases and Salts
Question 1.
Chemically, how can the elements found in nature be divided?
Answer:
1. In present times, 114 elements are known to us.
2. These elements combine in several ways and give rise to a very large number of compounds.
3. On the basis of their chemical properties aU the compounds can be added to three groups. They are:
- Acids,
- Bases and
- Salts.
4. Thus, all the compounds of this world belong to one or the other group.
5. In this sense, a compound may be acidic, basic or a salt.
![]()
Question 2.
What is an acid?
Answer:
Acids: An acid is a compound having hydrogen which when dissolved in water releases ie. dissociates hydrogen ions (H+) (to be specific (H3O+ ions).
Example:

Hydrochloric acid (HCl), sulphuric acid (H2SO4,) nitric acid (HNO3), etc. are all examples of acids.
(Note: Water H2O also has hydrogen in it but when you add water to water, it does not release hydrogen ions (H+). Hence, water is not acid. Similarly, NaOH has hydrogen but it does not release hydrogen ions (H+) when dissolved in water. NaOH is a base. So, only those substance which on dissolving to water release hydrogen ions (H+) are called acids.)
Testing of acid:
If you put acid on blue litmus paper, the blue litmus paper will turn red. This means the substance put is acid.
Question 3.
What is a base?
(a) Bases:
1. A base is a metal hydroxide substance which when dissolved in water release hydroxide (OH) ions. (Second definition: A metal hydroxide or a metal oxide substance when dissolved in acid produces salt and water and hence is called a base.)
2. Sodium hydroxide (NaOH), calcium hydroxide (Ca(OH)2, potassium hydroxide (KOH), Calcium oxide (CaO), etc. are bases

Testing of base:
If you put base on red litmus paper, the red litmus paper will turn blue. This means the substance put is base.
Question 4.
What is a salt?
Answer:
A salt is an ionic compound which is formed from the neutralization reaction of an acid and a base. Thus, we get salt when we react an acid with a base.
Note:
(1) When an acid and a base are mixed in right proportion, both acid and base lose their properties. In other words, they neutralize each other. Such a reaction is called neutralization reaction.
(2) When salt is dissolved in water it gets ionized into anions and cations)
![]()
Question 5.
State two ways to test if the substance is acid.
Whether a substance is an acid or not can be tested with any of the given methods:
Answer:
1. Litmus paper: If you place a drop of a given substance on a moist litmus paper and it turns red, then the substance is acid.
2. Test with metals: If you put metal in an aqueous substance and the reaction releases hydrogen gas then it means that the aqueous substance is acid.
Question 6.
What are olfactory indicators?
Answer:
1. Olfactory means ‘relating to the sense of smell’. Those substances whose smell changes in acidic or basic solutions are called olfactory indicators.
2. When an acid or a base is added to an olfactory substance, the characteristic smell of that substance cannot be detected.
Question 7.
How does onion and vanilla essence help in finding if a substance is acid or a base?
Answer:
1. If you add onion juice (or vanilla essence) to a base, the onion juice (or vanilla) will lose its smell. However, it will not lose the smell if added to acid.
2. Thus change in smell will help us to find out if the substance added to onion (or vanilla) is acidic or basic.
Question 8.
State three properties of acids and bases.
Answer:
Properties of acid:
- Acids are sour in taste
- They turn blue colour litmus paper into red
- Acids react with base and form salt and water
Properties of bases:
- Bases are bitter in taste
- They change red litmus paper to blue
- Bases react with acids to form salt and water
![]()
Question 9.
What happens when an acid reacts with metal? Give one example.
Answer:
When acid reacts with metal, metallic salt of that metal and hydrogen gas are produced.
Acid + Metal → Salt of Metal + Hydrogen gas
Example:
1. When zinc metal is added to sulphuric acid the reaction gives out zinc sulphate which is a salt and hydrogen gas.
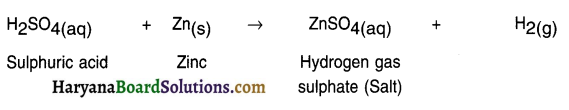
Question 10.
What happens when a base reacts with a metal? Give one example.
Answer:
When a strong base reacts with certain metals, it produces salt and hydrogen gas.
Base + Metal → Salt + Hydrogen gas
Example:
1. When sodium hydroxide (NaOH) reacts with certain metals like zinc Zn, salt and hydrogen gas are produced.

Question 11.
When zinc metal is treated with dilute HCl or dilute H2SO4, hydrogen gas is evolved, but with dilute HNO3 hydrogen gas is not evolved. Explain.
Answer:
Zinc metals reacts with dilute HCl and dilute H2SO4 evolving hydrogen gas. Since Zn metal is more reactive than H2 gas, Zn can displace H2 gas from dilute HCl and dilute H2SO4 solutions.
Reaction:
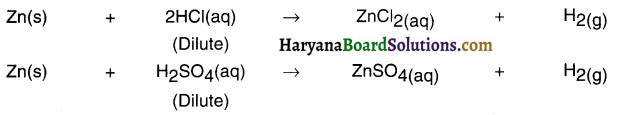
Hydrogen gas is not evolved by the reaction of Zn metal with dilute HNO3, because HNO3 is a strong oxidizing agent and H2 gas evolved during reaction is oxidized to H2O. Therefore, H2 gas is not obtained during the reaction of Zn with HNO3.
Question 12.
What happens when an acid reacts with metal carbonate or metal hydrogen carbonate? Give one example.
Answer:
Reaction of acid with metal carbonate or metal hydrogen carbonate:
When acids react with metal carbonate or metal hydrogen carbonate, most acids produce salt, water and carbon dioxide gas.

![]()
Question 13.
What happens when carbon dioxide in less proportion and excess proportion is passed through the solution of lime water? OR State the reactions that take place when carbon dioxide is passed through lime water (calcium hydroxide solution).
Answer:
On passing carbon dioxide in less proportion through lime water (calcium hydroxide solution), the solution turns milky because a white milky precipitate of calcium carbonate is formed.

On passing excess carbon dioxide through lime water, precipitate of calcium carbonate dissolves due to formation of soluble calcium bicarbonate.

Question 14.
What happens when an acid and a base react? Give one example.
Answer:
Reaction of acid with base:
- When acid reacts with base, salt and water are produced.
- Since base neutralizes the effect of acid, this reaction is called neutralization reaction.
Acid → Base → Salt + Water
Example:
When sodium hydroxide, a base reacts with hydrochloric acid, it produces salt and water.
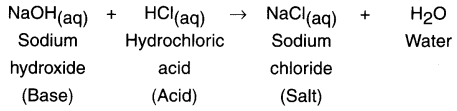
Question 15.
What is formed when copper oxide reacts with dilute hydrochloric acid? State the change in colour that you will observe along with the balanced chemical reaction. OR What happens when a metal oxide reacts with acid? Give one example.
Answer:
Reaction of acid with metal oxide:
1. When acid reacts with metal oxide, salt and water are produced.
Acid + Metal oxide → Salt + Water
2. When hydrochloric acid reacts with copper oxide, a salt of copper (Il) chloride is formed.
Example:
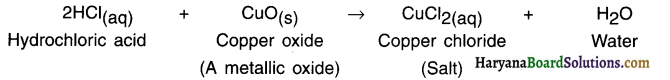
The colour of the solution is bluish-green due to the formation of copper chloride.
Question 16.
What happens when a non-metallic oxide reacts with a base?
Answer:
When a non-metallic oxide reacts with base, the reaction gives out salt and water.
Example:
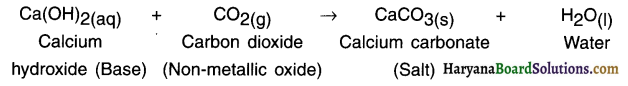
Question 17.
How are hydrogen ions produced when an acid is dissolved in water? Explain with necessary chemical equations. Also mention method of writing the hydrogen ions so formed.
Answer:
1. When an acid (or an acidic substance) dissolves in water, it produces hydrogen (H+) ions.
2. This happens because all acids contain positively charged hydrogen (H+) ions.
Example:

Thus, hydrogen ion must always be written as H+(aq) or hydronium ion (H3O+).
![]()
Question 18.
Write a brief note on base and alkali.
Answer:
Base:
- Chemical or substances which are hydroxides of metal and have a bitter taste are called bases.
- All metal oxides and metal hydroxide are bases.
- Fo example, sodium hydroxide (NaOH), calcium oxide (CaO), calcium hydroxide Ca(OH)2, etc. are bases.
- Washing soda (Na2CO3 10H3O), baking soda(NaHCO3), etc. are also bases or basic substances.
Alkali:
- Those bases which can dissolve in water are called alkalis.
- Sodium hydroxide (NaOH). potassium hydroxide (KOH), etc. are alkalis or say water soluble bases.
- When a base (or a basic substance) is dissolved in water, it always produces hydroxide (OH–) ions.
- Thus base is a substance which dissolves in water to produce hydroxide (OH–) ions in solution.
Example:

As shown in the above reaction, when sodium hydroxide which is a base, is dissolved in water it produces hydroxide (OH) ions over and above sodium (Na+) ions.

Here, when potassium hydroxide is dissolved in water, it gives hydroxide (OH) ions and potassium ions.
Question 19.
State the ions responsible for acidic and basic behaviour. Explain the acidic and basic behaviour by reaction with water.
Answer:
H+ or H3O+ ions in aqueous solution are responsible for acidic character and OH ions in an aqueous solution are responsible for basic character.
![]()
H+(aq) ions are formed in aqueous solution of HCl. So it can be said that HCl is an acid.
![]()
OH– ions are formed in an aqueous solution of NaOH. So NaOH is a base.
Question 20.
One should dissolve acid in water cautiously. Give reason. OR During dilution, acid must be added to water, not vice-versa.
Answer:
1. The process of dissolving an acid or a base is highly exothermic.
2. If one adds acid to water suddenly and in large amount or if one adds water to acid, the reaction will emit a lot of heat. This can even break the glass container.
3. The hot contents may come out of the container with an explosion and burn our eyes and body.
4. Hence, while making dilute solution of acid, acid must only be added to water that too slowly and by stirring continuously.
Question 21.
What Is dilution? How do you prepare a dilute acid?
Answer:
1. The process of mixing an acid or base with water decreases the concentration of ions (H3O/OH) per unit volume. The acid/base so formed is called dilute acid/base.
2. For making dilute acid one should slowly add concentrated acid in small amounts to water and stir continuously.
Question 22.
Write a short note on strong and weak acids.
Answer:
Strong acids:
1. An acid which gets completely ionized completely in water or say which completely dissociates in water and produce a large amount of hydrogen [H+] ions (or say hydronium [H3O+] ions) is called a strong acid.
2. Depending upon their structures, different acids produce different number of H+ ions. For example, 1 mole of hydrochloric acid will produce more hydrogen [H+] ions as compared to 1 mole acetic acid.
Weak acids:
1. An acid which does not ionize completely (i.e. does not dissociate completely in water) and thus produce a small amount of hydrogen [H+] ions (or say H3O+ ions) is called a weak acid.
2. For example, when acids such as acetic acid, lactic acid, citric acid, tartaric acid, etc. are dissolved in water, they do not completely ionize and so are called weak acids.
![]()
Question 23.
Write a short note on strong and weak bases.
Answer:
Strong base:
- A base, which completely ionizes in water and thus produces a large amount of hydroxide (OH) ions, is called a strong base or a strong alkali.
- For example, when sodium hydroxide (NaOH) and potassium hydroxide (KQH) are dissolved in water, they completely get ionized and so are called strong bases.
Weak base:
- A base, which does not ionize completely in water and thus produces a small amount of hydroxide (OH) ions, is called a weak base or a weak alkali.
- For example, ammonium hydroxide (NH4OH) and calcium hydroxide Ca(OH)2 when dissolved in water do not completely ionize in water and so are considered weak bases.
Question 24.
Explain the methods of measuring the strength of an acid or a base. Strength (or weakness) of an acid or a base can be measured through following methods:
Answer:
(a) Through universal indicator :
1. Universal indicator is a mixture of many different indicators or say dyes which when added to a solution, changes the colour of the solution and thus indicate its pH value.
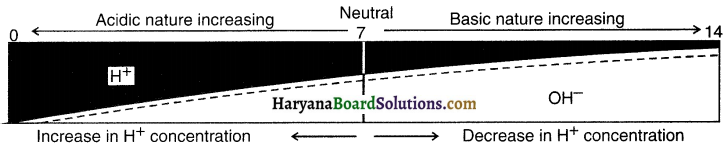
(b) Through pH scale:
1. To measure the acidic/basic nature of an aqueous solution, a scale called pH scale has been developed.
2. The pH scale measures concentration of hydrogen [H+] ions in the solution.
3. In German language, P of pH stands for ‘potenz’ i.e. power.
4. The scale points range from O to 14. 0 means very acidic and 14 means very alkaline. Scale point means neutral solution.
5. A lower pH value means there is higher concentration of H+ ions and hence the solution is acidic. So, as one moves towards O from 7, the solution goes on becoming more and more acidic.
6. Similarly, a higher pH value means there is lower concentration of H+ (and higher concentration of OH–) ions and hence the solution is basic. So, as one moves from 7 to 14, the basicity of the solution increases.
![]()
Question 25.
Write a note on pH scale.
Answer:
Through pH scale:
1. To measure the acidic/basic nature of an aqueous solution, a scale called pH scale has been developed.
2. The pH scale measures concentration of hydrogen [H+] ions in the solution.
3. In German language, P of pH stands for ‘potenz’ i.e. power.
4. The scale points range from O to 14. 0 means very acidic and 14 means very alkaline. Scale point means neutral solution.
5. A lower pH value means there is higher concentration of H+ ions and hence the solution is acidic. So, as one moves towards O from 7, the solution goes on becoming more and more acidic.
6. Similarly, a higher pH value means there is lower concentration of H+ (and higher concentration of OH–) ions and hence the solution is basic. So, as one moves from 7 to 14, the basicity of the solution increases.
Question 26.
How do you measure the strength of an acid or a base?
Answer:
1. The strength of an acid is determined by the number of H ions it produces, where as strength of a base is determined by the number of OH ions the base produces.
2. If we take 1 molar concentration (1 mole or 1 m acid dissolved in 1 litre of solution) of hydrochloric acid and acetic acid, then the acid which produces more number of H+ ions will be considered the stronger among the two. In this case, hydrochloric is a strong acid whereas acetic acid is weak.
3. Using the same method one can find the strength or weakness of a base. However, in base, the OH– ions are released and hence they are counted (instead of H+).
Question 27.
Discuss the importance of pH In daily life.
Answer:
Importance of pH In everyday life:
(1) Importance of pH in existence of living beings:
- The physiological reactions occurring in our body takes place between a narrow range of 7.0 to 7.8 pH. If due to any reason the pH range gets disturbed, we may face several problems in the body.
- Even other living beings cannot tolerate more changes in their pH level.
- For example, when acid rain (i.e. rain having pH level of 5.6) falls into water reservoirs like rivers, ponds, etc., it decreases pH level of these water bodies and makes them highly acidic.
- Under such circumstances, existences of aquatic organisms like fish, microorganisms and vegetation is risked.
(2) Importance of pH in soil:
- Shrubs grow well if the pH level of soil is maintained between 6.5 to 7.3.
- The soil having pH <6.5 is called acidic soil, and that having pH > 7.3 is called alkaline.
(3) Importance of pH indigestion of food:
- Stomach plays an important role in digesting food.
- When we eat food, stomach secretes hydrochloric acid, The pH of this acid ¡s between 1 and 3.
- At such a low value of pH, an enzyme called pepsin becomes active which helps in digesting food.
(4) Importance of pH in stopping tooth decay:
- The outer layer of the teeth is made up of calcium phosphate. This layer does not dissolve in water but gets decayed when pH of inner side of mouth becomes less than 5.5. This causes tooth decay.
- When we eat food, bacteria decomposes the food particles that remain in the mouth and produce acid. This decreases the pH in the mouth.
(5) Self-defence by animals and plats through chemical warfare:
- When a honey bee bites a human being, pain, irritation and swelling is felt at the site of bite.
- This occurs because the honey bee releases acidic poison into human body during the bite.
- To neutralize the effect of this acid to some extent, substances like baking soda are applied around the bite. Stinging hair of nestle leaves injects methanoic acid and causes burning pain.
Question 28.
Indigestion causes pain and irritation. Suggest how to cure this. OR How does baking soda help in relieving stomach pain and Irritation?
Answer:
1. During indigestion, the stomach produces excessive acid. This causes pain and irritation.
2. To neutralize the effect of the acid, bases must be used. Such bases are called antacids i.e. anti-acids.
3. Magnesium hydroxide (Milk of magnesia) is one such antacid. Baking soda is also a mild base which helps in neutralizing excess acid. (Note: Eno available in medical store is also one type of antacid.)
![]()
Question 29.
When tooth pH goes below 5.5 it leads to decay. How would you prevent it?
Answer:
1. When pH in the mouth goes below 5.5, the bacteria of the mouth start producing acids and decaying the teeth.
2. The best way to prevent this is to clean the mouth properly after eating.
3. Toothpastes are basic in nature. Hence, cleaning the teeth with tooth paste neutralizes excess acid and prevents tooth decay.
Question 30.
What is acid rain? How does it affect aquatic life?
Answer:
1. If the pH of rain water becomes less than 5.6, then such rain is called acid rain.
2. When such acid water flows in water bodies such as rivers, ponds and lakes, it increases the acidity of these water sources.
3. The living organisms including humans have quite a narrow pH band of 7.0 to 7.8. Hence, even slightest change in water source makes survival difficult.
Question 31.
Why distilled (pure) water is used as a solvent in laboratories ? How does distilled water self – ionise?
Answer:
1. Distilled (pure) water is neutral. So in laboratories a solution is made using distilled water in order to get a correct pH value.
2. When an acid or a base is added to distilled water, the solution produces hydronium (H3O+) and hydroxide (OH–) ions respectively through self ionization of water. The self-ionization reaction is as under:
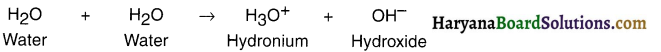
Question 32.
The aqueous solution of the salt produced by neutralization of weak acid and strong base possesses basic nature, while aqueous solution of salt produced by neutralization reaction of weak base and strong acid possess acidic nature – Explain.
Answer:
1. When the pH level of an aqueous solution increases more than 7, the solution goes on becoming basic.
2. In a neutralization reaction, when a weak acid and a strong base react, the salts obtained in the reaction hydrolyse to produce hydroxide [OH] ions.
3. As the level of [OH] ions increases in the solution, the solution becomes basic in nature.
4. In case, when in neutralization reaction, reaction takes place between a weak base and a strong acid, the salt obtained in the reaction hydrolyses in water to produce hydronium [H3O+] ions.
5. The increased level of [H3O+] ions in the aqueous solution makes the solution acidic.
6. Thus, the aqueous solution of the salt produced by neutralization reaction of weak acid and strong base possesses basic nature, while aqueous solution of salt produced by neutralization reaction of weak base and strong acid possess acidic nature.
Question 33.
What is neutralization reaction?
Answer:
Neutralization reaction:
1. When an acid and a base are mixed in right proportion, both acid and base lose their properties. Such a reaction is called neutralization reaction.
2. Thus, the reaction of acid and base is called a neutralization reaction. Moreover, reaction of acid and base gives us ‘salt’ and ‘water’.
Neutralization reaction can be generalized as follows:
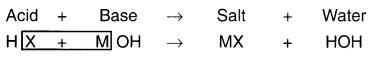
Where, H refers to hydrogen ion
OH refers to hydroxide ion
MX refers to Salt
HOH refers to water (H2O)
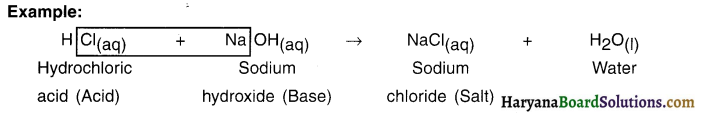
Question 34.
What s a salt?
Answer:
1. A salt is an ionic compound which is formed by the neutralization reaction of an acid and a base. Thus, we get salt when we react an acid with a base.
2. When we dissolve a salt in water it will get ionized and release cation (i.e. the positive +ve ion) and anion (i.e. the negative -ve ion).

3. ‘Salt’ is a general name and it does not refer only to NaCl i.e. sodium chloride. There exists a huge number of salts other than NaCl.
4. NaCl i.e. common salt so formed can be further used to produce several other products.
5. Sodium hydroxide (NaOH), baking soda (NaHCO3), washing soda (Na2CO3 10H2O), bleaching powder (CaOCl2) are all examples of salts.
Question 35.
How does a salt gets its name?
Answer:
The general form of naming a salt is ‘cation anion’. This means the name of cation (i.e. the positive +ve ion) of the salt will be put first and then name of its anion (i.e. the negative -ve ion) will be put second.
Example:
(a) NaCl: In NaCl salt, sodium Na+ has positive ions i.e. cations and chlorine CF has negative ions i.e. anions. Hence, we call NaCl salt as sodium chloride.
(b) K2SO4: Applying the same rule, the name of this salt is potassium sulphate.
![]()
Question 36.
What is a family of salts?
Answer:
1. There are several types of salts. The properties of all the salts are not completely different. In other words, properties of several salts are similar.
2. In general, salts having same type of cations (+ve ions) or anions (-ve) belong to the same family.
Example:
(a) Family of sodium salts (salts having Na ion): Na2SO4, NaCl, NaNO3, Na2CO3
(b) Family of chloride salts (salts having C ion): NaCl NH4Cl
(c) Family of sulphate salts (salts having SO4 ion): K2SO4, Na2SO4, Ca2SO4, MgSO4, CuSO4
Preparation of Important Salts
List of important salts that we will study in this section.
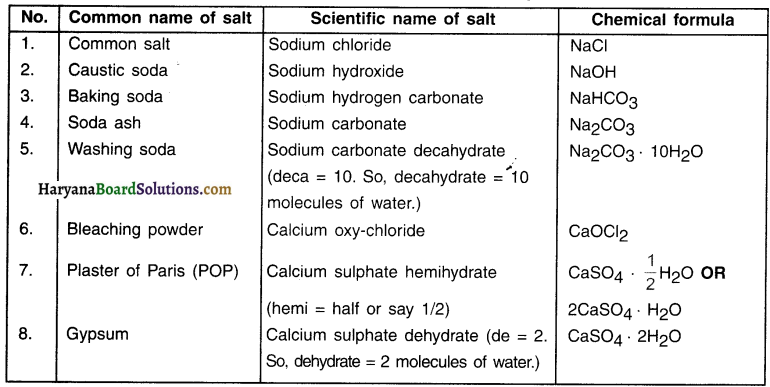
Question 37.
What is brine? State its two important uses.
Answer:
1. The concentrated solution of sodium chloride i.e. concentrated solution of NaCl + H2O is called brine.
2. Brine is used for preparing many compounds, however two products are prepared directly using brine.
They are:
- Caustic soda (sodium hydroxide) and
- Baking soda (sodium hydrogen carbonate).
Question 38.
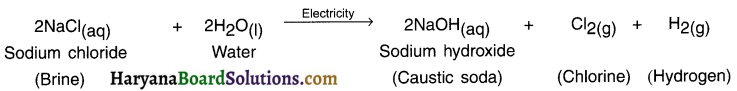
Write a note on causltc soda (sodium hydroxide). OR Write a note on chlor-alkali process. Chemical name of caustic soda: Sodium hydroxide
Answer:
Chemical formula: NaOH
Preparation:
1. When electricity is passed through brine, it gets decomposed and produces three products. They are:
- Sodium hydroxide,
- Chlorine gas and
- Hydrogen gas.
Reaction:
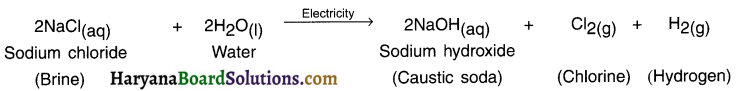
2. The chlorine gas is produced at anode (+ve electrode) and hydrogen at cathode. Sodium hydroxide is formed near the cathode.
3. The process of preparing NaOH is also called chlor-alkali process because the process gives out sodium hydroxide which is an alkali and chlorine.
![]()
Question 39.
State uses of the products formed in the chior-alkali process.
Answer:
Uses of products formed in chlor-aikali process:
- NaOH: Making soap and detergent, paper, artificial fibres, de-greasing metals, etc.
- Chlorine gas: To disinfect water, added in swimming pools, making PVC, CFCs and pesticides
- Hydrogen gas: As a fuel, for making fertilizers and in making margarine
Question 40.
Write a note on preparation of baking soda.
Answer:
Chemical name of baking soda: Sodium hydrogen carbonate
Chemical formula: NaHCO3
Preparation:
When brine reacts with ammonia in the presence of carbon dioxide gas, it produces sodium hydrogen carbonate and ammonium chloride.
Baking soda is a mild, non-corrosive basic salt.
Question 41.
Give an idea and preparation about soda ash. OR What happens when you heat baking soda? OR State and explain the reaction of baking soda when it Is heated during cooking. OR State the preparation of sodium carbonate.
Answer:
Chemical name of soda ash: Sodium carbonate
Chemical formula: Na2CO3
Preparation:
When baking soda gets heated during cooking, it produces sodium carbonate (soda ash) along with carbon dioxide and water.
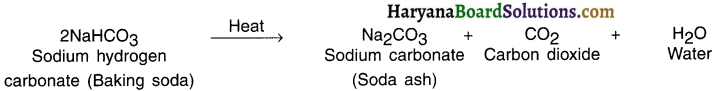
Question 42.
What happens when baking soda is heated or mixed with water? When baking soda is heated or mixed with water than following action takes place:
Answer:

Carbon dioxide produced during this reaction causes bread or cake to rise making them soft and spongy.
Question 43.
Mention uses of baking soda:
Answer:
1. Baking socia is a soda (or a salt) commonly used in kitchen for making cakes, crispy pakoras, khaman, etc.
2. It is used for making baking powder commonly used in kitchen.
3. It is also used in making antacids. Antacids cure acidity of stomach.
4. To make soda-acid fire extinguishers
5. To make several industrial products
![]()
Question 44.
How is washing soda prepared?
Answer:
Chemical name of washing soda: Sodium carbonate decahydrate
Chemical formula: Na2CO3 10H2O
Preparation:
When sodium carbonate (i.e. soda ash) is dissolved in water, and recrystallized it gives washing soda crystals containing 10 molecules of water of crystallization.

Washing soda is a basic salt.
Question 45.
What does the number 10 represents In the formula Na2CO3 10H2O i.e. the formula of washing soda?
Answer:
The number 10 in the formula Na2CO3 10H2O tells us that there are lo water molecules in washing soda. Hence, it is also called decahydrate.
Question 46.
State the uses of washing soda.
Answer:
Uses of washing soda:
- It is used in making glass and paper.
- It is used for making sodium based compounds such as borax.
- It is used as a cleaning agent such as washing powder and soap.
- For removing permanent hardness of water.
Question 47.
Write a note on preparation of bleaching powder and its uses.
Answer:
Chemical name of bleaching powder: Calcium oxy-chloride
Chemical formula: CaOCl2

Preparation:
1. It is prepared by the action of chlorine on slaked lime (Calcium hydroxide).
2. On passing chlorine gas over dry slaked lime (CaCOH2), bleaching powder is obtained.
Uses of bleaching powder:
- For bleaching cotton and linen clothes in textile industry, for bleaching wood pulp in paper-making factories and bleaching washed clothes in laundries.
- As an oxidizing agent in many chemical industries.
- For disinfecting drinking water to make it free of germs.
Question 48.
State the uses of bleaching powder.
Answer:
Uses of bleaching powder:
- For bleaching cotton and linen clothes in textile industry, for bleaching wood pulp in paper-making factories and bleaching washed clothes in laundries.
- As an oxidizing agent in many chemical industries.
- For disinfecting drinking water to make it free of germs.
Question 49.
What is plaster of Paris? State its chemical name and preparation.
Answer:
Chemical name of plaster of Paris (POP): Calcium sulphate hemihydrate (hemi = half or say 1/2)
Chemical formula: CaSO4 \(\frac{1}{2}\)H2O (OR 2CaSO4 . H2O)
Preparation:
CaSO 2H2O refers to a compound called gypsum. Its chemical name is calcium sulphate dehydrate (de =2).
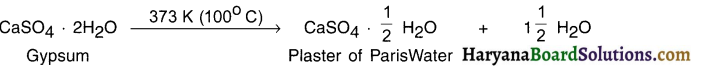
- On heating gypsum at 373 K, it loses water molecules and forms calcium sulphate hemihydrate (Plaster of Paris) and \(1 \frac{1}{2}\) molecule of water.
- In CaSO4 . \(\frac{1}{2}\)H2O, the half molecule of water is attached as water of crystallization.
- Plaster of Paris is a white powder. On adding water, it again turns to gypsum having hard solid mass.
Question 50.
What do doctors use to join the fractured bones?
Answer:
1. Doctors use white powder of plaster of Paris to join the fractured bones.
2. On adding water to this powder, it forms a paste which can be easily applied on the fractured area. When the paste becomes dry it becomes solid hard substance called gypsum.
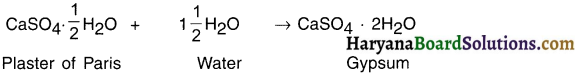
Question 51.
What is water of crystallization? Give examples.
Answer:
(a) The fix number of water molecules present in one formula unit of salt is called water of crystallization.
Examples:
(1) Calcium suphate hemihydrate (CaSO4. H2O) contains 1/2 i.e. half molecule of water of crystallization.
(2) Sodium carbonate decahydrate (Na2CO3 . 10H2O) contains 10 molecules of water of crystallization.
![]()
Question 52.
List out two differences between acids and base on the basis of their chemical properties.
Answer:
| Acid | Base |
| 1. When acids are dissolved in water they release (H+) ions. 2. Acid reacts with lime stone to liberate carbon dioxide gas. | 1. When bases are dissolved in water they release (OH–) ions. 2. Base reacts with oils to form soapy solution. |
Question 53.
State two difference between organic acid and mineral acid.
Answer:
| Organic acid | Mineral acid |
| 1. Acids present in plants and animais i.e. occurring naturally are called organic acids. Example: Citric acid, lactic acid, etc. are organic acids. | 1. Acids prepared from minerals found in earth are called mineral acids. Example: Hydrochloric acid, sulphuric acid, etc. are mineral acids. |
Question 54.
What will be the action of the following substances on litmus paper? Dry HCl gas, Moistened NH3 gas, Lemon juice, Carbonated soft drink, Curd, Soap solution.
Answer:
1. Dry HCl gas: No change on litmus paper
2. Moistened NH3 gas: Red litmus will turn blue.
3. Lemon juice: Blue litmus will turn red.
4. Carbonated soft drinks: They contain carbonic acid. So blue litmus will turn red.
5. Curd: It contains lactic acid. So, blue litmus will turn red.
6. Soap solution: It is basic. So, red litmus will turn blue.
Question 55.
Name the acid present in ant sting and give its chemical formula. Also give the common method to get relief from the discomfort caused by the ant sting.
Answer:
When ant stings, it releases formic acid (or methanoic acid) in our body. Its chemical formula is HCOOH. Mix some baking soda with water and apply it on the sting. Baking soda is basic and so it will neutralize the acidic effect and provide relief.
Question 56.
What happens when nitric acid is added to egg shell?
Answer:
Egg shells contain calcium carbonate (CaCO3). When nitric acid (HNO3) is added to it, it results in brisk effervescence due to the formation of CO2 gas. The reaction is as follows.
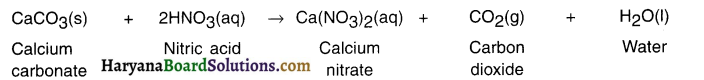
Question 57.
A student prepared solutions of (i) an acid and (ii) a base in two separate beakers. She forgot to label the solutions and litmus paper is not available In the laboratory. Since both the solutions are colourless, how will she distinguish between the two?
Answer:
Since the student does not have litmus, she can use any other indicator like methyl orange, phenolphthalein, etc. She can also use a natural indicator such as turmeric.
![]()
Question 58.
When zinc metal is treated with a dilute solution of a strong acid, a gas s evolved, which is utilized in the hydrogenation of oil. Name the gas evolved. Write the chemical equation of the reaction involved and also write a test to detect the gas formed.
Answer:
When zinc reacts with dilute solution of strong acid (like hydrochloric acid HCl), it forms salt and hydrogen gas is evolved which is used in hydrogenation of oil.

To test the presence of H2 gas take a burning candle near the mouth of the test tube. The gas burns with a pop sound indicating presence of hydrogen gas.
Question 59.
How would you distinguish between baking powder and washing soda by heating?
Answer:
On heating baking soda (NaHCO3), carbon dioxide (CO2) gas is produced. It turns lime water milky.

If you heat washing soda (Na2CO3 . 10H2O), water of crystallization is given out and the sail becomes anhydrous. The presence of water of crystallization given as product can be tested b treating it with anhydrous CuSO4. It is white coloured which turns blue when it comes in contact ol anhydrous CuSO4.
![]()
Question 60.
Salt A commonly used in bakery products on heating gets converted Into another salt B Which itself Is used for removal of hardness of water and a gas C is evolved. The gas C when passed through lime water, turns It milky. Identify A, B and C.
Answer:
Salt A: It is used in bakery and also gives Na2CO3 . Hence it is sodium bicarbonate NaHCO3.
Salt B: It is sodium carbonate Na2CO3 because it is used for removal of hardness of water.
Gas C: It turns lime water milky and hence gas C is carbon dioxide CO2.

Question 61.
A compound X of sodium is commonly used In kitchen for making crispy pakoras. It is also used for curing acidity in the stomach. Identify X. What is its chemical formula? State the reaction which takes place when it is heated during cooking.
Answer:
Compound X: Sodium bicarbonate or sodium hydrogen carbonate or baking soda
Formula: NaHCO3
Chemical reaction:
![]()
Question 62.
For making cake, baking powder is taken. If at home your mother uses baking soda instead of baking powder in cake,
(a) How will it affect the taste of the cake and why?
(b) How can baking soda be converted into baking powder?
(c) What is the role of tartaric acid added to baking soda?
Answer:
(a) Baking powder contains tartaric acid. This acid reacts with sodium carbonate (Na2CO3) produced during decomposition of NaHCO3 and neutralizes it.
If only sodium hydrogen carbonate (baking soda) is used in making cake, then sodium carbonate formed from it by the action of heat (during baking) will give a bitter taste to cake.
(b) By adding tartaric acid to baking soda we can convert baking soda into baking powder.
(c) Tartaric acid neutralizes the sodium carbonate formed during decomposition of NaHCO3. Hence, it makes the cake tasty and prevents it from becoming bitter in taste.
Question 63.
“The aqueous solution of the salt produced by neutralization of weak acid and strong base possesses basic nature, while aqueous solution of salt produced by neutralization of weak base and strong acid possesses acidic nature.” Explain.
Answer:
1.
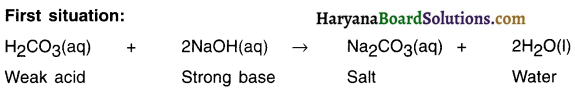
2. When we react carbonic acid which is a weak acid with a strong base of sodium hydroxide what we get is sodium carbonate.
3. The aqueous solution of sodium carbonate contains higher concentration of OH ions. Hence, the solution possesses basic nature.
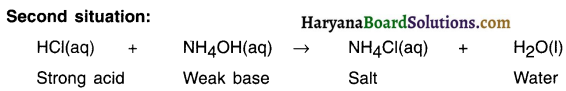
4. When we react hydrochloric acid which is a strong acid with a weak base, we get ammonium chloride.
5. This aqueous solution contains higher concentration of H+(aq) ions compared to the concentration of OH+ (aq) ions. Therefore solution possesses acidic nature.
Question 64.
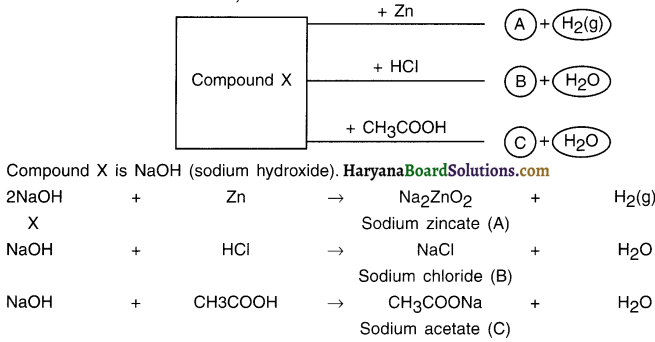
Identify compound X on the basis of the reactions given below. Also, write the name and chemical formulae of A, B and C.
Answer:
Question 65.
While eating food, you spill some curry on your white shirt. You immediately scrub It with soap.
What happens to its yellow colour on scrubbing with soap? Why? What happens to this stain. when the shirt is washed with plenty of water?
Answer:
When we scrub the shirt with soap, its colour changes from yellow to reddish brown. This happens because soap is basic in nature and the colour of turmeric changes from yellow to reddish brown in basic medium. If we wash the shirt with plenty of water, the stain will again turn yellow.
Very Short Answer Type Questions
Question 1.
How can we broadly classify all the compounds known to us?
Answer:
All the compounds that exist can be classified as
- Acids,
- Bases or
- Salts.
![]()
Question 2.
What are acids? Name two acids.
Answer:
Substances which when dissolved in water release hydrogen ions (H+) are called acids. Hydrochloric acid and sulphuric acid are two common acids.
Question 3.
State two properties of acids.
Answer:
Acids are sour in taste, Acids conduct electricity when dissolved in water, Acids react with metals to form salt and hydrogen gas.
Question 4.
What are bases?
Answer:
Bases are hydroxides of metals which give hydroxide (OH–) ions when mixed with water.
Question 5.
State two properties of bases.
Answer:
Bases have a bitter taste. They change red litmus paper to blue.
Question 6.
What is salt?
Answer:
A salt is an ionic compound which is formed from the neutralization reaction of an acid and a base. Thus, we get salt when we react an acid with a base.
Question 7.
Read the paragraph and point out the erroneous statement.
Answer:
Acids are sour and they release H+ ions. Bases are bitter and they released H3O+ ions. Also, bases turn litmus paper blue. Litmus is an artificial indicator to test presence of acids.
- Bases are sour
- Litmus is natural
- Bases release H+ ions Statement (i) and (iii) are erroneous.
Question 8.
Name two natural and synthetic indicators.
Answer:
1. Natural: Litmus and turmeric,
2. Synthetic: Methyl orange and phenolphthalein.
Question 9.
A knife, which is used to cut a fruit, was immediately dipped into water containing drops of blue litmus solution. If the colour of the solution changes to red, what inference can be drawn about the fruit and why?
Answer:
Since the colour of blue litmus turned red, the fruit is acidic.
![]()
Question 10.
What is an olfactory indicator?
Answer:
A substance whose smell changes in acidic or basic solution is called an olfactory indicator.
Question 11.
Name two olfactory indicators.
Answer:
Onion and vanilla extract.
Question 12.
What is a concentrated acid?
Answer:
An acid that contains minimum amount of water is called a concentrated acid.
Question 13.
What is a dilute acid?
Answer:
An acid obtained by mixing concentrated acid with large amount of water is called a dilute acid.
Question 14.
Name a substance that does not contain hydroxide ion yet it acts as a base. Also give its formula.
Answer:
Ammonia (NH3)
Question 15.
You are given gold, silver and platinum. Which of these will not react with acid easily? Why?
Answer:
None. Because all are noble metals.
Question 16.
What will happen if you add zinc granules to dilute sulphuric acid?
Answer:
Bubbles containing H2 gas will be formed on zinc granules.
Question 17.
Rasika took some dilute sulphuric acid in a test tube and added a few pieces of zinc granules to it. Her friend Shyam was supposed to write the observation in the journal but he was absent that day. As a science student write the observation that must have taken place in this experiment.
Answer:
We can observe that bubbles form on the surface of zinc granules indicating formation of gas. When we pass this gas through the soap solution, the gas gets trapped in soap molecules and bubbles are formed. The gas is hydrogen and it burns with a pop sound when a burning candle is brought near it.
Question 18.
What will be produced when you pass carbon dioxide gas through lime water?
Answer:
A milky precipitate of calcium carbonate (CaCO3).
Question 19.
Take a small amount of copper oxide in a beaker and add dilute hydrochloric acid in it. State the chemical reaction.
Answer:
CuO + 2HCl CuCl2 + H2O
Question 20.
You might have seen lemon or tamarind juice being used to clean tarnished surface of copper vessels. Explain why these sour substances are effective in cleaning the vessels?
Answer:
Copper is metal whereas lemon juice and tamarind are acidic. When acid reacts with metal oxides, salt and water is formed. This makes the metal vessels shiny again
Question 21.
Why lemonade should not be prepared in copper vessel?
Answer:
Lemon is highly acidic and reacts vigorously with copper metal and causes copper poisoning. Hence………..
![]()
Question 22.
Take about 0.5 g of sodium carbonate (Na2CO3) in test tube A and about 0.5 g of sodium hydrogencarbonate (NaHCO3) in test tube B. Add about 2 mL of dilute HCl in both the test tubes. State the observation of this experiment.
Answer:
When acids react with metal carbonates and metal hydrogen carbonates, they produce salt, water and carbon dioxide gas. This happens in both the test-tubes.
Question 23.
Look at the reaction given below and mention what will happen if you pass excess carbon dioxide from it. Also state the reaction.
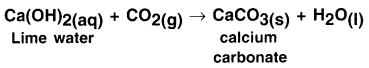
Answer:
On passing excess carbon dioxide through the given solution, precipitate of calcium carbonate dissolves due to formation of soluble calcium ‘ bicarbonate.
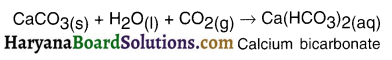
Question 24.
What is neutralization reaction?
Answer:
When acid and base mix in right proportions, both of them lose their properties and produce salt and water. Such a reaction is called neutralization : reaction.
Question 25.
What will be the colour of solution if you add two drops of phenolphthalein solution in dilute NaOH solution?
Answer:
The solution will become pink.
Question 26.
Why does the colour of phenolphthalein
Answer:
NaOH gets neutralized by acid. Hence, the colour of phenolphthalein changes.
Question 27.
Give the neutralization reaction of potassium hydroxide with sulphuric acid.
Answer:
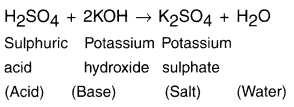
Question 28.
Write one word for the following:
(a) Water soluble base
(b) A substance which dissociates on dissolving in water to produce hydroxide ions.
Answer:
(a) Alkali
(b) Base
Question 29.
Write one word for the following:
(a) A substance which dissociates on dissolving in water to produce hydrogen ions.
(b) A reaction between an acid and a base to form salt and water.
Answer:
(a) Acid,
(b) neutralization
Question 30.
Give reason in one sentence: ‘Solution of sulphuric acid conducts electricity whereas alcohol does not’.
Answer:
Solution of sulphuric acid contains charged ions H+ and SO4-2 which helps in conducting electricity whereas alcohol does not. Hence,………..
Question 31.
Although compounds like alcohol and glucose contain hydrogen, they are not acids. Why?
Answer:
Although alcohol and glucose contain hydrogen, they do not give hydrogen ions in water and hence are not categorized as acids.
Question 32.
![]() What is happening in this reaction?
What is happening in this reaction?
Answer:
Here, when potassium hydroxide, a base is subjected to water, it generates hydroxide (OH–) ions. Which out of the solutions of glucose,
Question 33.
Which out of the solutions of glucose, alcohol, hydrochloric acid, sulphuric acid and sodium hydroxide, will not conduct electricity?
Answer:
Glucose and alcohol will not conduct electricity because they will not release ions.
![]()
Question 34.
What will happen it you add water to strong acid for producing a dilute acid?
Answer:
A lot of heat will be produced. This may splash out the acid and burn our bodies. Even the glass container may break.
Question 35.
What is dilution?
Answer:
Mixing acid or base with water results in decrease in the concentration of H3O+ or OH ions per unit volume. Such a process ¡s called dilution and the acid/base is called diluted.
Question 36.
An experiment was done in which about 1g solid NaCl was taken in a clean and dry test tube. Then some concentrated sulphuric acid was added to the test tube. The reaction produced hydrochloric acid. On the basis of the above activity, what do you infer about the acidic character of : (i) Dry HCl gas, (ii) HCl solution?
Answer:
The experiment suggests that hydrochloric acid produces hydrogen ions in the presence of water. But, dry hydrochloric acid does not release hydrogen ions. Thus, only HCl solution is acidic whereas dry HCl is not.
Question 37.
What is a universal Indicator?
Answer:
An indicator which can pass through a series of colour changes over a wide range of H3O+ ion concentration is called universal indicator. It is a mixture of several indicators.
Question 38.
What is pH scale?
Answer:
A scale that measures the concentration of hydrogen ion in a solution is called a pH scale.
Question 39.
Arrange the following in an increasing order of their pH values: NaOH solution blood, lemon juice.
Answer:
NaOH < Blood < Lemon juice
Question 40.
Why 1M HCl solution will have a higher concentration of H ions compared to 1M CH3COOH solution?
Answer:
1. HCl i.e. hydrochloric acid is a strong acid. Hence, HCl molecules dissociate completely into H+ ions and Cl– ions and produce more H+ ions.
2. CH3COOH i.e. acetic acid is a weak acid and so it does not dissociate completely. Hence, it produces less H+ ions. As a result
Question 41.
A student added a few drops of liquid P into distilled water. He observed that the pH of the water decreased. Can you guess the pH of the liquid P?
Answer:
We know that pH of distilled water is 7. On adding liquid P, the pH of the water decreased which means that P could be any acid such as HCl or H3SO4, etc.
Question 42.
Which type of substances are taken for getting relief from acidity?
Answer:
Basic substances or say antacids
Question 43.
Separate the following acids into strong acids and weak acids. Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
Answer:
Strong acid: Hydrochloric acid, nitric acid and sulphuric acid:
Weak acid: Citric acid acetic acid and formic acid
Question 44.
What do you mean by family of salts?
Answer:
1. There are several types of salts. The properties of all the salts are not completely different, In other words, properties of several salts are similar.
2. In general, salts having same type of cations (+ve ions) or anions (-ve) belong to the same family.
![]()
Question 45.
Which salt do you obtain by the reaction of hydrochloric acid and sodium hydroxide?
Answer:
Sodium chloride (NaCl)
Question 46.
Name two salts that you can directly prepare from brine.
Answer:
(1) Caustic soda (sodium hydroxide) and
(2) Baking soda (sodium hydrogen carbonate).
Question 47.
State chemical reaction for preparing caustic soda.
Answer:
Reaction:
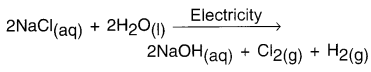
Question 48.
What is chlor-alkali process?
Answer:
When electricity is passed through an aqueous solution of sodium chloride, it decomposes to form sodium hydroxide and chlorine gas. This process is called chlor-alkali process.
Question 49.
How does the soda-acid fire-extinguisher extinguish the fire?
Answer:
Soda-acid fir-extinguisher extinguishes the fire by stopping the contact of air with fire.
Question 50.
How is bleaching powder produced? State is reaction.
Answer:
Bleaching powder is produced by the action of chlorine on dry slaked lime (Calcium hydroxide)
Ca(OH)2 + Cl2 → CaOCl + H2O
Question 51.
State two uses of bleaching powder.
Answer:
(a) For bleaching cotton and linen in textile industry.
(b) For disinfecting drinking water.
Question 52.
What is baking powder?
Answer:
Baking powder is a mixture of baking soda and a mild edible acid such as tartaric acid.
Question 53.
Why baking soda is used in making antacid?
Answer:
Baking soda is alkaline and so it neutralizes excess acid in the stomach and provides relief from acidity. Hence, it is …………..
Question 54.
Recrystallization of sodium carbonate gives washing soda. State the reaction.
Answer:
Na2CO3 + 10H2O → Na2CO3 10H2O
![]()
Question 55.
State two uses of washing soda.
Answer:
(a) To prepare glass and soap,
(b) To remove permanent hardness of water
Question 56.
State two uses of POP.
Answer:
(a) Doctors use POP for setting fractured bones.
(b) It is used for making false and decorative ceilings.
Fill in the Blanks:
1. When NaOH is added to a cloth strip treated with onion extracts, the onion smell ……………………..
Answer:
Cannot be detected OR Vanishes.
2. Cloth strip treated with onion + dilute. NaOH solution = …………………… (state observation)
Answer:
Onion smell will not be detected in the cloth and the cloth will change to green colour.
3. H+ ions cannot separate from HCl molecules in the absence of …………..
Answer:
Water
4. For a neutralization reaction, H X + M OH → ………………..
Answer:
MX + HOH
5. Universal indicator is used for …………………
Answer:
Obtaining approximate pH of a solution.
6. In pH scale, scale points O’ = and
Answer:
O = very acidic, 14 = very basic
7. Generally, with the universal indicator is used to measure pH.
Answer:
Paper impregnated
8. Higher the hydronium ion concentration, is the pH value.
Answer:
Lower
9. If OH– > 10-7, solution will be
Answer:
Basic
10. If a red litmus paper is dipped into a solution and it turns blue, then it can be said that the solution has pH range between
Answer:
7and 14
11. In order to have good growth and development of shrubs, the soil should have pH
Answer:
Near 7
12. Bacteria present in the mouth produce base by degradation of food particles left in the mouth after eating.
Answer:
False
13. Two substances that have almost equal neutral pH are ………………..
Answer:
Blood and water
![]()
14. The pH values of aqueous solutions A, B, C and D are 2.9, 3.5, 1.6 and 4.2 respectively. The correct order of their acidic strength is …………………….
Answer:
C>A> B>D
15. …………….. decreases the pH inside the mouth.
Answer:
Acids
16. Aqueous solution of is applied around the place of bite, to get relief from the effect of bite of honey bee.
Answer:
Baking soda
17. The sting of nettle leaves inject (name of the compound) in the body.
Answer:
4 Methanoic acid
18. The other name and formula of calcium sulphate hemihydrate …………..
Answer:
Plaster of Paris : CaSO4 \(\frac{1}{2}\)H2O.
True Or False
1. The solution which has no effect on any litmus paper is neutral. — True
2. Tartaric acid is stronger than nitric acid. — False
3. As a thumb rule, all organic acids are weak acids and mineral acids are strong acids. — True
4. H3O+ = OH– = 10-7 — True
5. Sodium hydroxide and magnesium hydroxide are basic and hence work quite well in curing stomach acidity. — False
6. Hydrogen ions cannot exist alone. — True
7. HCl solution is acidic but its dry form is not. — True
8. Dissolution of acid (or base) in water releases is an endothermic process. — False
9. Limestone, marble and chalk are forms of calcium carbonate. — True
10. The pH of a neutral solution is O. — False
11. The pH of gastric juices is about 1.2. — False
12. You need to heat gypsum at 378 K for forming calcium sulphate hemihydrate. — False
13. Carbon dioxide gas is mainly responsible for making the cake soft. — True
14. If we make the crystals moist, we can see blue colour of copper sulphate reappearing. — True
Match the Following
1. Match the acids given in Column (A) with their correct source given in Column (B)
Column (A) | Column (B) |
| (a) Lactic acid (b) Acetic acid (c) Citric acid (d) Oxalic acid | (i) Tomato |
Answer:
(a-4) (b-3) (c-2) (d-1)
![]()
2. Match the important chemicals given in Column (A) with the chemical formulae given in Column (B)
| Column (A) | Column (B) |
| (a) Plaster of Paris (b) Gypsum (C) Bleaching Powder (d) Slaked Lime | (i) Ca(OH)2 (ii) CaSO4 (iii) CaSO4 1/2 H2O (iv) CaOCl2 . 2H2O |
Answer:
(a-2) (b-3) (c-4) (d-1)